July 7, 2020
BIOINVEST NEWS
BIOINVEST BREAKING NEWS — Special Update — NEW RECOMMENDATION: Vaxart (VXRT) – VXRT’s Oral Vaccine for COVID-19 Poised to Enter Human Testing
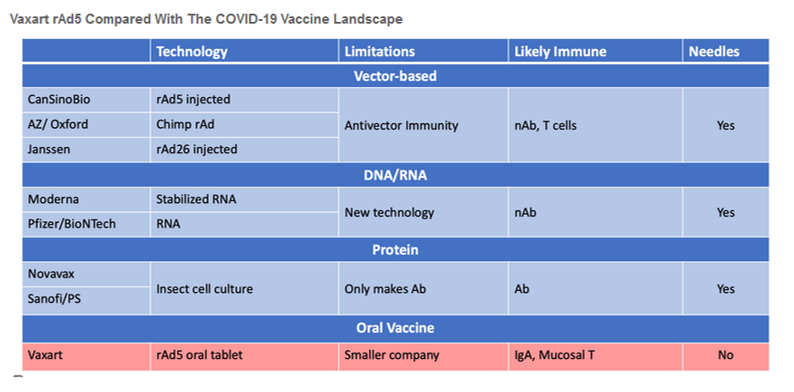
We are initiating coverage of Vaxart with a BUY rating (VXRT $7.88, Market Capitalization $748 million). With proprietary vaccine technology that has recently been validated by being selected to participate in the government’s Operation Warp Speed (OWS) program (https://www.hhs.gov/about/news/2020/06/16/fact-sheet-explaining-operation-warp-speed.html), VXRT is about to begin Phase I human testing of an oral (pill) vaccine for SARS-CoV-2, the coronavirus that causes COVID-19. In our view, the ability for a vaccine to provide both mucosal (IgA) and systemic immunity could be a game changer for preventing infection from COVID-19. COVID-19 is a respiratory disease that is primarily transmitted by viral particles that enter through the mucosa – nose, mouth or eyes – strongly suggesting that mucosal immunity could serve as the first line of defense. The ability to stimulate both mucosal and systemic immunity with a pill provides two significant advantages to injectable vaccines that only generate systemic immunity.

VAAST is the name of Vaxart’s platform vaccine technology and has been published in at least half a dozen medical journals since 2012 (see above). Vaxart uses a specific virus called adenovirus type 5 (Ad5) as part of its novel VAAST™ platform to help train the immune system to recognize and defeat dangerous invading pathogens. Vaxart recombinantly removes the DNA Ad5 uses for viral replication, so the virus cannot make copies of itself. Vaxart then uses the disabled Ad5 virus as a delivery system, or “vector,” to carry the genes coding for the “antigen,” a component of the target virus that is known to generate a protective immune response, and the “adjuvant,” a molecule that stimulates the innate immune system. The Ad5 vector delivers these two genes to the epithelial cells lining the mucosa of the small bowel.
When the altered Ad5 virus enters a cell and delivers the genes for the antigen and the adjuvant, the genes are expressed, and the cell manufactures the vaccine antigen for the immune system to recognize, as well as the adjuvant that stimulates the immune system to mount a targeted response against the vaccine antigen. These two molecules “teach” the immune system to recognize the antigen, a specific component of the pathogen. The next time the same antigen appears in the body, as part of a new infection, the immune system is prepared to mount a rapid and overwhelming response.
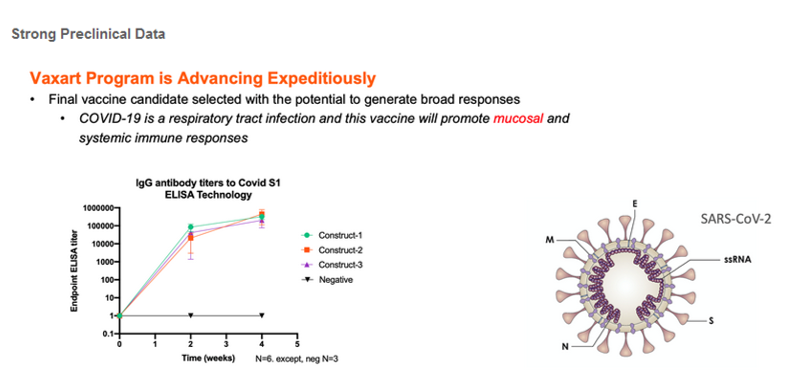
In May, VXRT’s oral COVID-19 vaccine, rAd5, was selected to participate in a non-human primate (NHP) challenge study, organized and funded by Operation Warp Speed, the new national program aiming to provide substantial quantities of safe, effective vaccine for Americans by January 2021. As an upstart to the COVID landscape, OWS has provided significant scientific validation for VXRT’s cutting edge oral vaccine technology. Earlier this year, the Company presented impressive preclinical data for rAd5 generating impressive immune responses that helped formed the the basis for the vaccine’s acceptance into Operation Warp Speed (see below). Like several vaccines selected for this program (e.g., AZN/Oxford, NVAX), we expect that VXRT will receive substantial government funding in the near-term for this program.
Manufacturing Capacity Established With Emergent, Kindred & AMS
Also last month, VXRT signed a partnership with Attwill Medical Solutions Sterilflow, LP (AMS) as a major resource for lyophilization development and large scale manufacturing including tableting and enteric coating for Vaxart’s oral COVID-19 vaccine. The partnership will enable the production of a billion or more COVID-19 vaccine doses per year. AMS will be assigning dedicated resources and equipment for the scale up and commercial production of the vaccine upon entering a formal agreement. The addition of AMS to existing VXRT CDMO partners (Emergent, Kindred) will “enable the large scale manufacturing and ultimate supply of our COVID-19 vaccine for the US, Europe and other countries in need.”
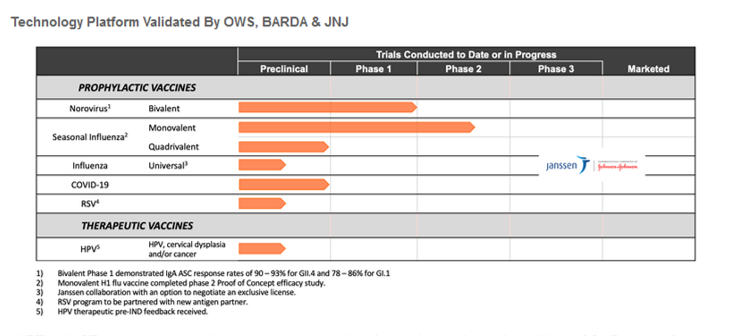
VXRT’s VAAST technology has delivered several oral vaccines for respiratory viruses in addition to COVID, such influenza, norovirus, RSV and a therapeutic vaccines starting with HPV. The Company has received ~$16 million in BARDA funding for an influenza vaccines, and a year ago Vaxart entered into a research collaboration agreement with Janssen Vaccines & Prevention B.V. (JNJ) to evaluate Vaxart’s proprietary oral vaccine platform for the Janssen universal influenza vaccine program (see above). In addition to the OWS inclusion, we believe these early deal help validate the VAAST technology. In fact, in our view, there is a chance that the Company eventually enters into a corporate collaboration for the COVID vaccine program.
New CEO Has Deal Experience
VXRT has new management as they appointed Andrei Floroiu as Chief Executive Officer in June. Mr. Floroiu is a highly experienced biopharma executive with a proven track record of value creation, with substantial financial, strategic and operational experience in the vaccine and biopharmaceutical industry. Mr. Floroiu was most recently with Agenus (AGEN), an immuno-oncology and vaccine pioneer, where he held executive positions and was responsible for structuring strategic partnerships and other transactions, including the origination and execution of a $115 million royalty transaction based on GSK’s Shingrix vaccine.
Upcoming VXRT Catalysts
- Raise capital, as of March 31, the company has $30 million in cash like many biotech companies in the current strong biotech financing environment, they are likely to raised capital. This will also be an opportunity to bring institutional investors into the shareholder list.
- File IND with FDA for COVID-19 Vaccine
- Start Phase I dose ranging trial in humans this summer
- Report Phase I/II Flu data
- Operation Warp Speed funding
Background
Vaxart is a clinical-stage biotechnology company developing a range of oral recombinant vaccines based on its proprietary delivery platform. Vaxart vaccines are administered using convenient room temperature-stable tablets that can be stored and shipped without refrigeration and eliminate the risk of needle-stick injury. Vaxart’s proprietary tablet vaccine delivery platform is suitable to deliver recombinant vaccines, positioning the company to develop oral versions of currently marketed vaccines and to design recombinant vaccines for new indications. Its development programs currently include tablet vaccines designed to protect against coronavirus, norovirus, seasonal influenza and respiratory syncytial virus (RSV), as well as a therapeutic vaccine for human papillomavirus (HPV), Vaxart’s first immuno-oncology indication. Vaxart has filed broad domestic and international patents covering its proprietary technology and creations for oral vaccination using adenovirus and TLR3 agonists.
Summary – There’s A New Kid In Town
In our view, VXRT is the leader in developing an oral vaccine for COVID-19. The ability to stimulate both mucosal and systemic immunity with a pill provides two significant advantages to injectable vaccines that only generate systemic immunity. The Company is poised for a string of significant catalysts once they raise money. Currently, there is little or no Wall Street research coverage on VXRT but we believe that will change soon as rAd5 gains further visibility via clinical trials and government funding.
We are recommending VXRT as a BUY under 15 with a TARGET PRICE of 30
Portfolio Transactions: We will buy $250K of VXRT in both the Model and Trader’s Portfolios.