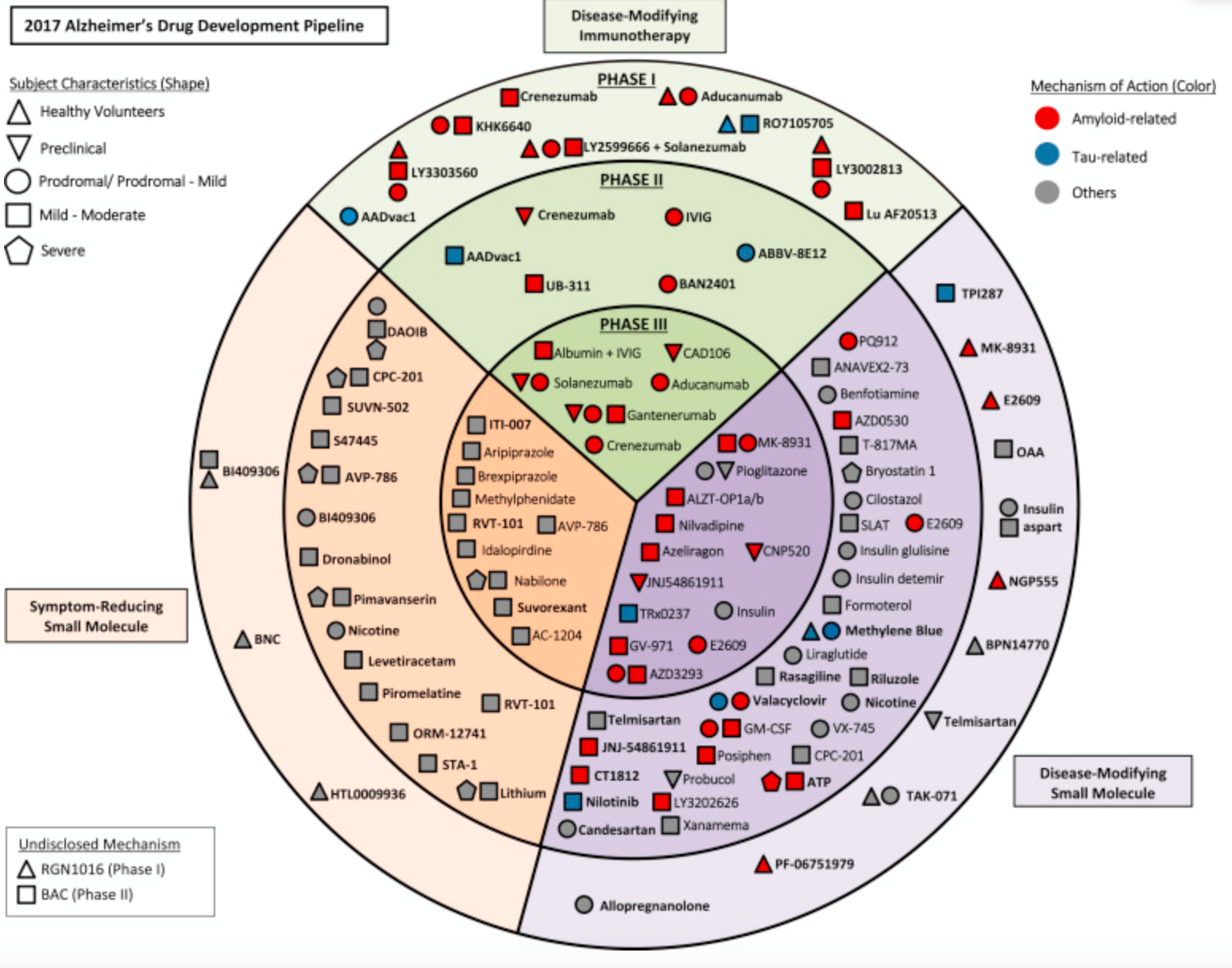
Two neurology scientific meetings have taken place this Fall and focused on hot new targets for neurological drug development- notably, beta-amyloid and tau. The CNS and Neurodegenerative Targets symposium was held in Boston last month (http://www.discoveryontarget.com/CNS-Neurodegenerative-Targets/) and then recently the American Neurological Association conference (https://2017.myana.org) included sessions on next-generation targets for Alzheimer’s (AD) and other brain diseases. While the clinical development inhibitors of BACE1 (beta-secretase 1 or beta-site amyloid precursor protein cleaving enzyme 1) are ahead of the tau compounds, there have been some notable failures aimed at that protein (e.g., MRK’s verubecestat back in 2012 and more recently, LLY’s solanezumab).
While beta-amyloid plays an important role in the pathophysiology of AD, data is emerging that the tau protein is potentially more critical in clinical intervention than BACE1, and/or some combination therapy of the two might be even better than either alone (see below). Tau-directed therapeutic approaches are a rapidly evolving field in translational Alzheimer’s research. Albeit still early in the clinic, the mechanisms of pathological tau assembly appear the most rational AD treatment targets at present. Controversy exists about intracellular versus extracellular treatment targets.
MTSL Recommended stocks IONS and SGMO have novel tau programs under development that in our view have yet to be recognized by investors as additional valuable components of each respective Company’s platforms. In this whitepaper, we point to recent and upcoming catalysts regarding both of these programs.
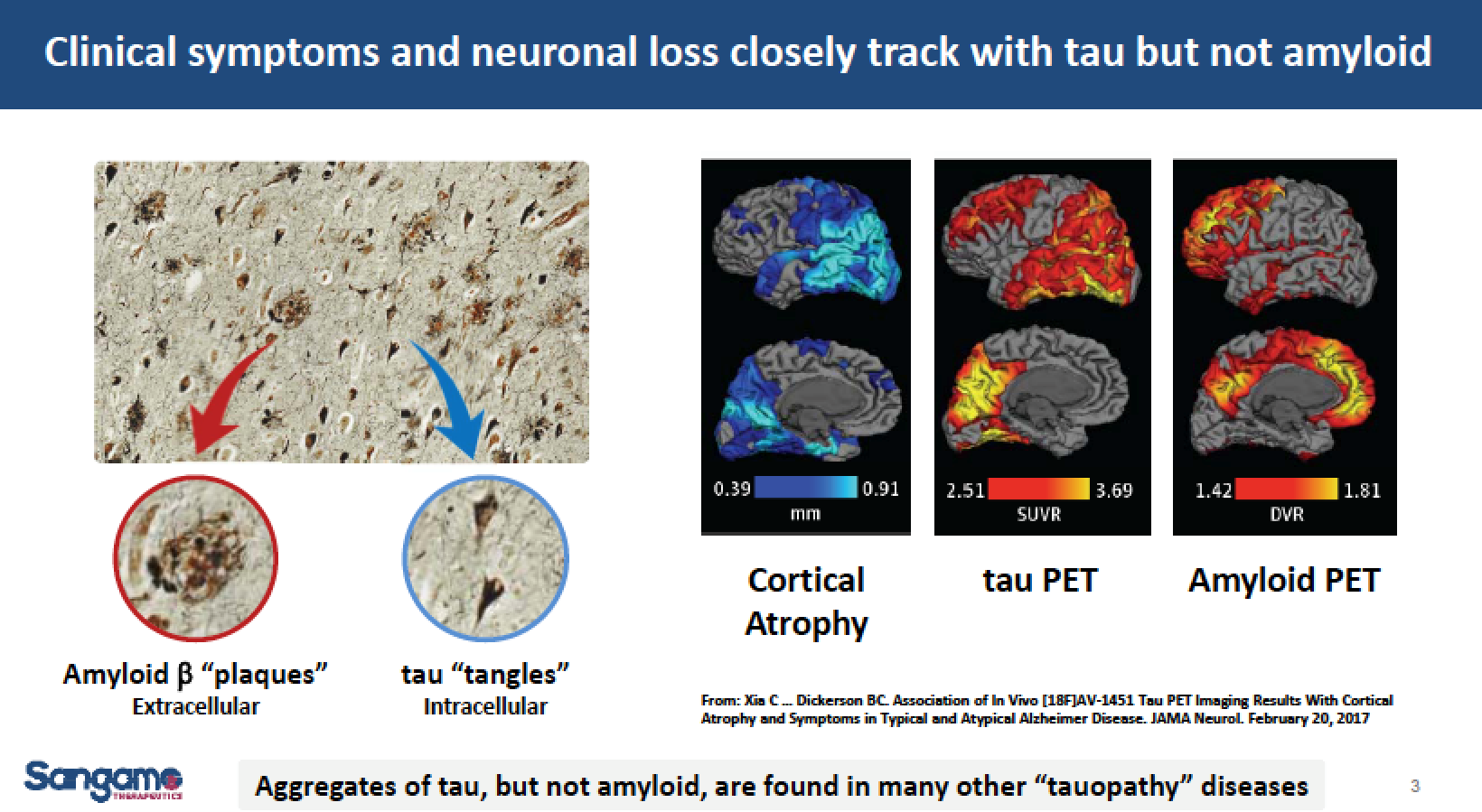
Tau misfolding (“tau tanglesâ€) leads to the accumulation of neurofibrillary tangles inside brain cells. These tangles are a hallmark feature of a spectrum of neurodegenerative diseases known as tauopathies, which include Alzheimer’s Disease (AD) and some forms of frontotemporal dementia (FTD). AD is the most common form of dementia, accounting for an estimated 70% of cases. FTD is a rare form of early-onset dementia resulting from neuronal damage in the frontal and temporal lobes of the brain. In contrast to amyloid plaques that may begin to deposit in the brain for up to 20 years before the onset of AD, tau deposits are spatially and temporally associated with the brain regions where atrophy occurs and neurocognitive deficits originate.
Biogen’s leadership in its MS franchise and novel approaches to AD research has led to its understanding the role of tau protein in neurodegenerative diseases and has resulted in a portfolio targeting two potential mechanisms. One approach involves tau spreading, which might cause neuronal death. BIIB092 is an antibody recently licensed from BMY (see BIIB/BMY deal below) that might address this problem by targeting extracellular tau and preventing its spread, thus slowing disease progression. ABBV, Roche/Genentch, Lilly, J&J, and others also have monoclonal antibody or vaccine programs targeting specific forms of tau.
The next technology involves stopping too much tau from being accumulated within the cell body, which could potentially be addressed by an antisense oligonucleotide – and the lead compound is from BIIB’s neurology IONS collaboration – IONIX-MAPTR. BIIB has previously noted that both approaches, extra cellular via a MAb and extracellular via antisense may ultimately end up being needed to effectively target tau.
IONIS-MAPTRx
IONIS-MAPTRx is designed to reduce the production of all of the many forms of tau in all regions of the brain. On Friday, October 13th, IONS initiated a Phase I/IIa clinical study of IONIS-MAPTRx in patients with mild Alzheimer’s disease (AD). IONIS-MAPTRx is an antisense drug designed to selectively reduce the production of microtubule-associated protein tau (MAPT), or tau protein, in the brain.
The three-month randomized, placebo-controlled, dose escalation Phase I/IIa clinical study will evaluate the safety and activity of IONIS-MAPTRx in approximately 44 patients with mild AD. In the study, IONIS-MAPTRx is administered as a once-monthly intrathecal injection directly into the cerebral spinal fluid, similar to the way SPINRAZA® (nusinersen) is administered. Biogen has an option to develop and commercialize IONIS-MAPTRx. In starting the trial, Ionis earned a $10 million milestone payment from partner BIIB.
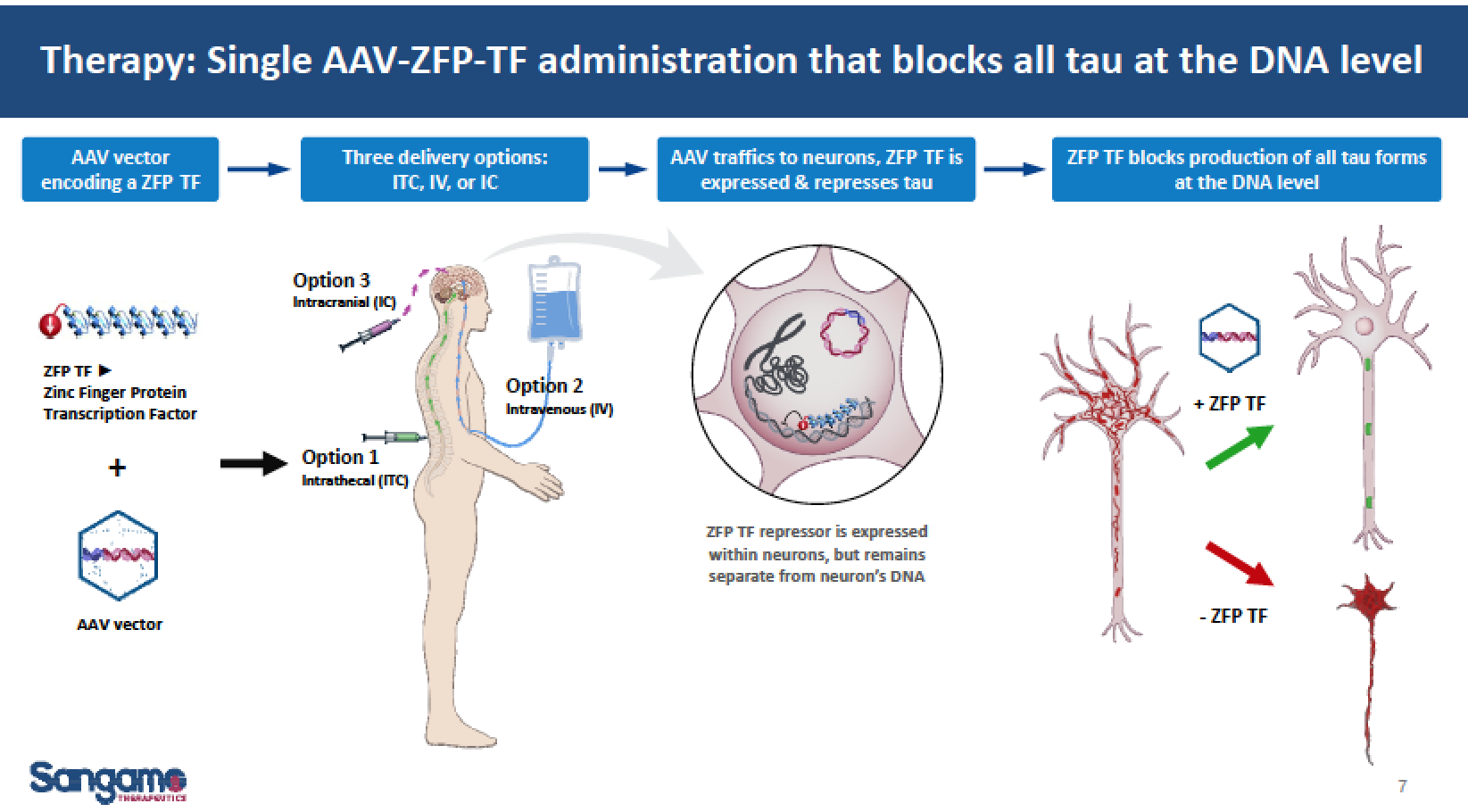
A third approach is the gene therapy of reducing mRNA MAPT tau protein expression with a single injection by SGMO. IONS has recently started human trials while SGMO presented very promising pre-clinical data earlier this year and expects to sign a partner for its tau program in the near future. We are unaware of anyone else other than SGMO targeting total tau with a single administration.
SGMO Anti-Tau ZFP
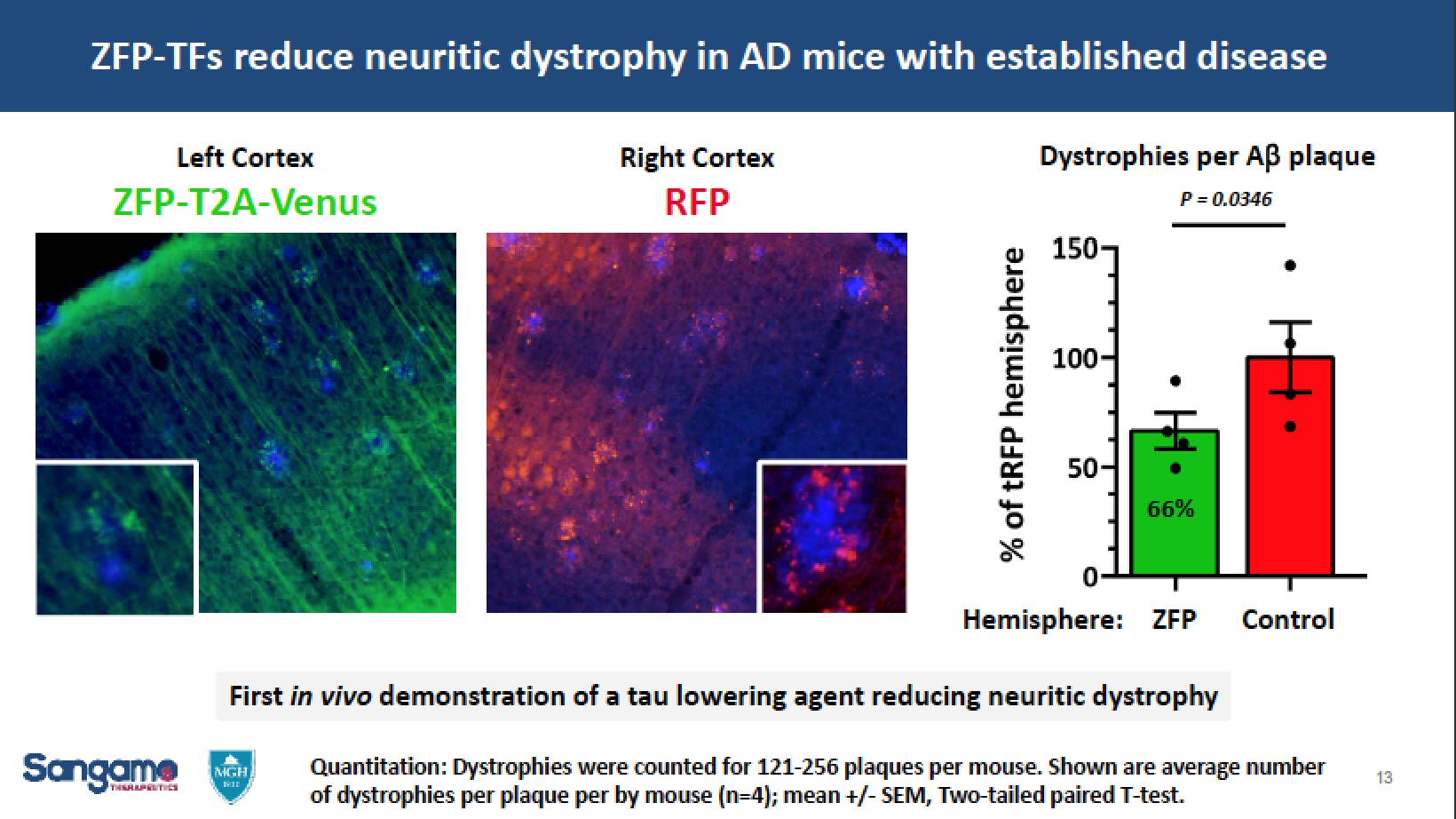
In March, SGMO scientists and researchers at Harvard Medical School presented new human in vitro and animal model data demonstrating significant reduction of tau mRNA and tau protein expression using the Company’s zinc finger protein transcription factor (ZFP-TF)-mediated gene regulation technology. These results are the first evidence of a tau-lowering agent demonstrating efficacy on neuritic dystrophy in an amyloid mouse model of Alzheimer’s disease. The data were presented by SGMO’s collaborators from Massachusetts General Hospital Alzheimer’s Disease Research Center and Harvard Medical School at the 13th International Conference on Alzheimer’s & Parkinson’s Diseases.
The March 29th presentation included data from in vitro studies conducted using induced pluripotent stem cell (iPSC) derived neurons demonstrating that a single administration of ZFP-TFs, via adeno-associated virus (AAV) vectors, resulted in greater than 90% reduction of human tau mRNA and protein. These data were consistent with a separate in vitro study using cortical neurons from wild-type mice, in which a greater than 90% reduction in tau mRNA and protein was achieved. Furthermore, specificity and off-target analysis in ZFP-TF-treated primary neurons revealed that tau was the only gene suppressed out of more than 26,000 coding transcripts analyzed.
Additionally, data from in vivo studies of wild-type mice demonstrated greater than 80% reduction of tau mRNA and protein in the hippocampus, as well as sustained and well-tolerated ZFP-TF expression with minimal impact on inflammatory markers. Finally, data from in vivo studies in an amyloid mouse model of Alzheimer’s disease demonstrated significantly reduced neuritic dystrophy after a single administration of ZFP-TFs in mice with established disease pathology.
The unknowns of BACE1 inhibitors include a) substrates; b) off-target toxicity and c) inhibition level.
Drugs to block this enzyme (BACE inhibitors) in theory would prevent the buildup of beta-amyloid and (per the Amyloid hypothesis) may help slow or stop Alzheimers disease. One outstanding question for BACE1 inhibitors discussed at the conference was how safe is BACE1 inhibition? Germline BACE1 knockout mice are found to display a variety of subtle and complex neurological phenotypes (i.e., memory deficits, seizure, axon guidance defects, hyperactivity, hypomyelination, and muscle spindle reduction), which are mediated by substrates other than APP (Jag1, CHL1, NRG1, VEGFR1, etc.), effectively raising the concerns around BACE1 inhibitors. A conditional whole body BACE1 knockout mouse model was created as a closer recapitulation of BACE1 inhibition in adult patients. Some of this background information is taken from a rather useful summary of the meeting from Andrew Fein at Wainwright & Co.
Although BACE1 inhibitors have shown to be relatively safe in adults (>4 years on drug in verubecestat EPOCH trial), at the meeting Dr. Robert Vassar (NorthWestern) expressed concern against BACE1 over-inhibition since adult neurons express BACE1 and a myriad of BACE1 substrates exist, pointing to potential side effects of BACE1 approach in the clinical setting. He suggested that caution is warranted when considering the downside of trials of BACE1 inhibitors (i.e., Merck’s verubecestat and Astrazeneca/Eli Lilly’s AZD3293) given that BACE1 inhibition is most likely to work in the preventative/prodromal stage of AD, which implies chronic use (and maybe even before symptoms set in) and commands a high safety hurdle. The level of BACE1 inhibition (or a ‘sweet spot’) required for therapeutic purpose (sufficient to flatten AD progression curve but low enough to mitigate untoward toxicity, if possible) remains an open question. To mitigate BACE1 inhibitor off-target toxicity, better biomarkers for the substrates (other than APP) are needed as more novel substrates are identified.
BACE1 dysregulation model for dystrophic axons in AD: an approach to stabilize microtubules. Previous studies showed that BACE1 levels are increased in dystrophic axons and terminals that surround plagues in APP Tg and AD brains. Additionally, microtubules are disrupted in dystrophic axon proximal to plaque but remain intact distally. A feed-forward mechanism is proposed by Dr. Robert Vassar’s lab that Abeta promotes BACE1 and β-secretase cleavage, which in turn elevates Abeta production. The accumulation of Abeta leads to subsequent degeneration of neurons (parallel to tau as a direct trigger). This is partially supported by the fact that Aβ42 oligomer treatment leads to microtubule disruption and BACE1 accumulation in primary hippocampal neurons (unpublished data), pointing to an Abeta-mediated mechanism of axonal dystrophy. Interestingly, Taxol (a microtubule stabilizer) treatment showed to rescue the neuronal toxicity (microtubule disruption) associated with Abeta. We note that microtubule disruption is an essential step in the neurodegenerative process as it triggers dystrophic axons, leads to deficiency of synaptic function, and eventually results in synaptic/neuronal loss and cognitive deficits. By breaking the vicious cycle of Abeta (or tau) induced neuronal degeneration, microtubule stabilizers may present as a promising therapeutic class (e.g., Cortice’s tau stabilizer TPI 287 is in Phase I trial). This approach may have a narrow therapeutic window as over-stabilization could lead to various problems.
Tau targeting via uptake pathway in a ‘prion-like’ model of tauopathies.
In the world of Abeta cascade hypothesis, Abeta is being called the ‘trigger’ and tau as the ‘bullet’ that kills neurons. Targeting tau pathways, in our view, may yield promising results from the mechanistic point of view (as the ‘bullet’ that kills neurons) and from the biomarker angle (better correlation with cognitive decline), although the temporalspatial spreading of tau proteins is not fully understood (and reliant on tau diagnostic tools). The ‘prion-like’ hypothesis of tauopathies suggest that tau travel behaves like prion proteins. Tau released from dysfunctional neurons enters healthy neurons by an uptake process. As tau aggregates in the neurons, a cascade of cell processes ensue which leads to neuronal dystrophy.
The pharmaceutical strategy, therefore, includes blocking the uptake of tau. Current pre- and clinical work is focusing on targeting tau aggregation via inhibitors, vaccines, and immunotherapy. Two publications elucidate the intracellular internalization of tau monomers and oligomers (oligomer is the toxic form of tau) and the critical role of heparin sulfate proteoglycans (HSPGs) in mediating tau aggregate binding, update and subsequent seeding of normal intracellular tau. Syndecan 4 (SDC4, a HSPG, which is increased in AD post-mortem prefrontal cortex) and 3-O-sulfation (brain specific) emerged as promising modifiers of tau internalization as the knock down/inhibition of both targets reduced tau uptake in hiPSC neurons in a mice model. Tau spreading in AD brains is being seen as a characteristic signature for disease progression, an intervention that inhibits the process may prove successful.
BIIB/BMS BMS-986168
In April, BIIB announced an agreement to exclusively license BMS-986168, a Phase II-ready antibody with potential in Alzheimer’s disease (AD) and progressive supranuclear palsy (PSP), from Bristol-Myers Squibb.
BMS-986168 is an antibody targeting extracellular tau, the protein that forms the deposits, or tangles, in the brain associated with AD and other neurodegenerative tauopathies such as PSP. PSP is a rare and devastating condition that affects movement, speech, vision, and cognitive function. Biogen plans to initiate Phase II studies for BMS-986168 in both AD and PSP.
Under the agreement, Biogen received worldwide rights to BMS-986168. Biogen will be responsible for the full development and global commercialization of BMS-986168 in AD and PSP. Bristol-Myers Squibb was paid $300 million upfront from BIIB and may receive up to $410 million for additional milestone payments and potential royalties. Biogen will also assume all remaining obligations to the former stockholders of iPierian related to BMY’s acquisition of the company in 2014. Biogen may pay up to $550 million in remaining milestones plus royalties including a near term $60 million milestone.
The Large Potential Of A SGMO Tau Deal Re: BMY/Ipierian
To put a potential SGMO tau deal in perspective, in April 2014 BMS acquired closely-held iPierian for $175 million, with the potential for additional development and regulatory milestone payments totaling $550 million, along with future royalties on net sales. With the recent SGMO/PFE deal for hemophilia A in May (i.e., $70 million upftont + ~$475 million in milestones, etc.), we believe a tau deal could be sizable since a) the AD markets dwarf that of even a big one like hem A; b) SGMO management has shown adeptness at creating high value deals; c) tau is a very hot protein and the Company has shown impressive preclinical data with >90% reduction in tau mRNA levels and d) the gene therapy space is white hot right now. On top of the $175 million BMY paid for Iperian, BIIB just gave BMY $300 million upfront for the same tau antibody plus another $410 million in milestone/royalties. Moreover, financially BIIB took on the BMY-Ipierian obligation of up to $550 million in milestones/royalties. So the total amount of the deals for BMS-986168 alone adds up to $475 million upfront plus $960 million in milestones plus future royalties on net sales. Since SGMO is still in preclinical development, a SGMO-tau deal is unlikely to be as large as the total BIIB/BMY/Ipierian amounts, but should nonetheless be sizable and remind investors that SGMO has a broad, deep and increasingly valuable platform.
IONS is a BUY under 55 with a TARGET PRICE of 75.
SGMO is a BUY under 16 with a TARGET PRICE of 20.