November 5, 2021
BIOINVEST NEWS: Precigen (PGEN)
R&D Update Highlights Depth & Breadth of Novel Pipeline – BUY
Precigen held a virtual R&D presentation yesterday, updating data on five clinical trials from the novel Ultra-CAR-T and AdenoVerse platforms. The company has delivered objective responses in late-stage, refractory cancer patients (AML, ovarian, HPV-positive tumors) utilizing their Ultra-CAR-T system plus which offer the fastest (overnight vs. 4-6 weeks) low-cost CAR-T therapies around. With the Adenovector platform, a number of HPV cancers/conditions may be treated safely with mono and/or combination therapy. The breadth of the platforms, in our view, is unique and exceptional. Reiterate BUY.
PRGN-3006 UltraCAR-T for AML – A dose-dependent expansion and persistence in both the non-lymphodepletion and the lymphodepletion cohorts was observed. An ORR of 50% (3 out of 6) was reported in the lymphodepletion cohort (Cohort 2) in patients treated at the two lowest dose levels. This included an ORR of 33% (1 out of 3) at Dose Level 1 and 67% (2 out of 3) at Dose Level 2. Objective responses included one partial response (PR) in a patient with extramedullary AML, one complete response with incomplete hematologic recovery (CRi) which was bridged to allo-HSCT, and one complete response with hematologic recovery (CRh).
Upcoming presentation: An abstract for the PRGN-3006 Phase 1 trial (Abstract# 825) titled, “Phase 1/1b Safety Study of PRGN-3006 UltraCAR-T in Patients with Relapsed or Refractory CD33-Positive Acute Myeloid Leukemia and Higher Risk Myelodysplastic Syndromes,” was selected for oral presentation at the 63rd ASH Annual Meeting on December 13, 2021 at 5:00 PM ET.
PRGN-3005 UltraCAR-T for Ovarian Cancer- Ten heavily pre-treated ovarian cancer patients have been enrolled. New data continue to show a favorable safety profile with no DLTs, no neurotoxicity, and no CRS reported. Data show dose-dependent expansion and persistence in the peripheral blood for more than 3 months after PRGN-3005 treatment without lymphodepletion, and clinical activity as evidenced by a decrease or stabilization of total target tumor burden at the first restaging in a majority of patients.
Next steps for -3005: Complete dose escalation in the IP and IV arms and, subsequently, incorporate lymphodepletion prior to PRGN-3005 infusion, which was cleared by the FDA. Additionally, based on the favorable safety profile and not using a viral vector the potential for repeat dosing is being evaluated.
PRGN-3007 Next Generation UltraCAR-T with Intrinsic PD-1 Inhibition – One of the most exciting new programs, PRGN-3007 is based on the next generation of UltraCAR-T platform. It is an investigational multigenic, autologous CAR-T cell therapy engineered to simultaneously express a CAR targeting receptor tyrosine kinase-like orphan receptor 1 (ROR1), mbIL15, a kill switch, and a novel mechanism for the intrinsic blockade of PD-1 gene expression. ROR1 is aberrantly expressed in multiple hematological and solid tumors with minimal expression in healthy adult tissues.
As announced last week, the FDA cleared the IND application to initiate a Phase 1/1b open-label trial designed to evaluate the safety and efficacy of PRGN-3007 in patients with advanced ROR1+ hematological (Arm 1) and solid (Arm 2) tumors. The target patient population for Arm 1 includes r/r chronic lymphocytic leukemia (CLL), r/r mantle cell leukemia (MCL), r/r acute lymphoblastic leukemia (ALL), and r/r diffuse large B-cell lymphoma (DLBCL). The target patient population for Arm 2 includes locally advanced unresectable or metastatic histologically confirmed triple negative breast cancer (TNBC). The trial will enroll in two parts: an initial 3+3 dose escalation in each arm followed by a dose expansion at the maximum tolerated dose. Arm 1 and Arm 2 will enroll in parallel.
Preclinical data: An abstract highlighting PRGN-3007 preclinical data (Abstract# 1694) titled, “Preclinical evaluation of PRGN-3007, a non-viral, multigenic, autologous ROR1 UltraCAR-T® cell therapy with novel mechanism of intrinsic PD-1 blockade for treatment of hematological and solid cancers,” will be presented as a poster presentation at the 63rd ASH Annual Meeting.
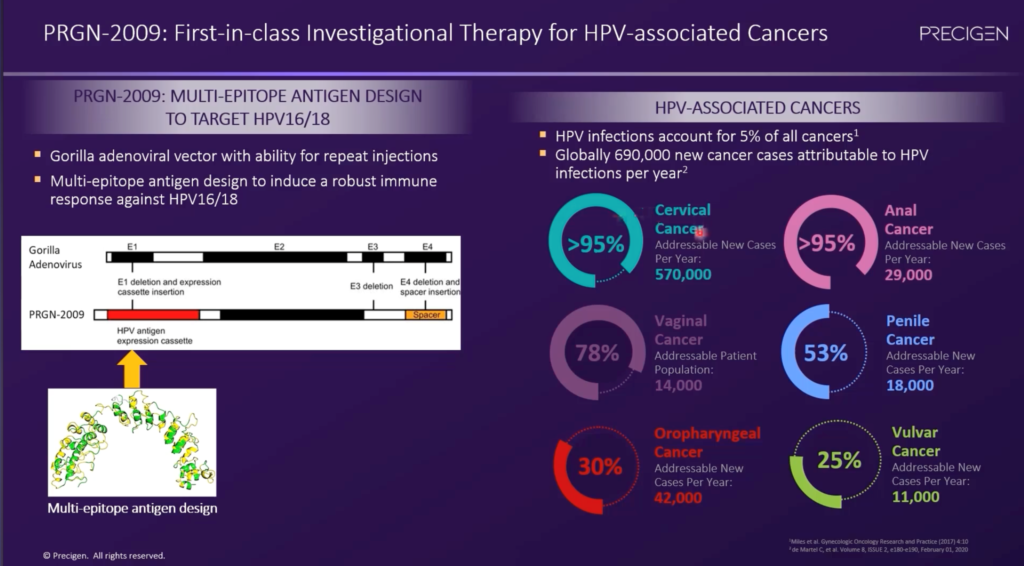
PRGN-2009 OTS AdenoVerse Immunotherapy for HPV-Positive Tumors – PRGN-2009 is an immunotherapy designed to activate the immune system to recognize and target HPV-positive solid tumors. PRGN-2009 is currently in a Phase 1/2 clinical trial under a CRADA with the NCI. The Phase 1 trial is evaluating safety and response of PRGN-2009 as monotherapy (Arm A) and in combination with M7824, Merck Serano’s novel bi-functional anti PD-L1/TGFbeta trap fusion protein (Arm B) in previously treated patients with recurrent or metastatic HPV-associated cancers. In the Phase 1 monotherapy arm, a DCR of 50% (3 out of 6 with stable disease (SD)) at the first restaging was observed. This includes a patient with durable (>1 year) SD who has received 16 PRGN-2009 monotherapy administrations. In the Phase 1 combination therapy arm, an ORR of 40% (2 out of 5) per RECIST v1.1 was observed. Objective responses included one ongoing CR at approximately 6 months after treatment initiation and one ongoing PR at approximately 7 months after treatment initiation. Additionally, a DCR of 60% (3 out of 5) at first restaging was observed.
PRGN-2012 OTS AdenoVerse Immunotherapy – PRGN-2012 is an investigational OTS AdenoVerse immunotherapy designed to elicit immune responses directed against cells infected with HPV 6 or HPV 11 for treatment of recurrent respiratory papillomatosis (RRP). PRGN-2012 is currently under evaluation in a Phase 1 clinical trial with the NCI. The Phase 1 trial is designed to follow 3+3 dose escalation of PRGN-2012 as an adjuvant immunotherapy following standard-of-care surgical removal of visible papillomas in adult patients with RRP. PRGN-2012 has been granted Orphan Drug Designation in patients with RRP. Preliminary data from three RRP patient case studies demonstrate very encouraging clinical activity of PRGN-2012 with reduction or elimination in the need for surgical interventions at the most recent follow-up, up to 12 weeks after PRGN-2012 treatment, compared to the recent history of surgical interventions for these patients before enrolling in the trial.
There are a slew of potential cancers that may be treated with PGEN’s UltraCAR-T and Adneovirus Immunotherapy platform technologies. Several of these indications may be eligible for rapid regulatory pathways, as there are no current options for a handful of these very late-stage indications. In addition, with wholly-owned PGEN platforms, the potential for corporate licensing deals and/or an outright acquisition in our view, is rising. UltraCAR-T is truly novel as it provides both time- and live-saving and yes, money saving treatments for late-stage, refractory cancer patients. AdenoVerse has broad implications in blockbuster markets in HPV-positive cancers and we are excited about the potential for the PRGN-2009 combo data. Several of these programs will be highlighted at the upcoming ASH meeting (December 11-14, https://www.hematology.org/meetings/annual-meeting). PGEN remains one of our favorite names.