MTSL Issue 957
IN THIS ISSUE: Delta Rampages
Since Last Issue: BTK: 4.8%; NBI: 9.1%; XBI: 3.2%; Model Portfolio: 5.1%; Trader’s Portfolio: 9.1%
SENTIMENT — The Wheel is Turning…
DELTA CONTINUES TO RAMPAGE WORLD, WHO ASKS FOR BOOSTER MORATORIUM, STATE DEPT./CDC ISSUE TRAVEL WARNINGS, DELTA ACCOUNTS FOR 93% IN U.S, EU BUYS 200 MILLION NOVAVAX DOSES
The World Health Organization called on Wednesday for a moratorium on coronavirus vaccine booster shots until the end of September, so that vaccine supplies can be focused on helping all countries vaccinate at least 10 percent of their populations. The agency made its appeal to the world’s wealthiest nations to address the wide disparities in vaccination rates around the world.
The U.S. State Department has listed the following countries and advised against traveling to them because of COVID-19; Andorra, Lesotho, Greece, Ireland, Malta, Kazakhstan, Curacao, French West Indies, Iran, Libya, the Marshall Islands, Syria and the Democratic Republic of Congo. The State Department issues a travel advisory for each country around the world and countries are given a ranking in four categories, including “exercise normal precautions,” “exercise increased caution,” “reconsider travel” and “do not travel.” The Centers for Disease Control and Prevention also issues travel advisories. There are currently 70 countries — about 35% of the world — it recommends avoiding traveling to because of COVID-19. If you must travel to any of the listed countries, the CDC recommends you get vaccinated first.
The latest data from the CDC shows that the DELTA variant accounts for at least 93% of all sequenced coronavirus in the U.S. For the two-week period ending July 31, all the different lineages of the DELTA variant made up an estimated 93% of cases that were sequenced. In some parts of the country with low vaccination rates, especially the Midwest region that includes Kansas, Iowa and Missouri, the percentages are even higher. Vaccination rates remain uneven across the U.S. with only about half of all eligible people nationwide fully vaccinated.
CDC Leak is Good & Bad News
The good news is the CDC report says that vaccinated individuals have a 25-fold reduction in risk of hospitalization or death compared to unvaccinated individuals, as well as an eight-fold reduction in disease, which translates to approximately 88% vaccine effectiveness against symptomatic infection. The bad news is the report stated the delta variant is more transmissible than MERS, SARS, Ebola, the common cold, the seasonal flu and smallpox. Another negative is that the variant is more likely to break through vaccine protection than other strains.
Daily Trends in COVID-19 Cases in the United States Reported to CDC
The European Commission has approved a supply contract with Novavax for the purchase of up to 200 million of its potential COVID-19 vaccine. Under the contract, EU states will be able to buy up to 100 million doses of the Novavax vaccine, with an option for 100 million additional doses until 2023, once the shot has been approved by the EU which is currently reviewing it.
White House Focuses on Life Sciences and Health
In a major departure from previous administrations, the White House Office of Science and Technology Policy — under the leadership of Eric Lander — will have an entire wing focused on life sciences and health. Other White House science offices have had heavy investments in physics, energy policy, and national security — in a reflection of the topics that were most important in the 1970s when the office was first created. In building the new wing, Lander and the Biden administration are signaling a broader push for high-risk, high-reward biomedical research. Lander has already begun poaching top scientists from other government agencies, including the NIH’s associate director of science policy, Carrie Wolinetz.
Acquisitions
Amgen said that it will pay up to $1.6 billion to buy TeneoBio, a privately held biotechnology company that specializes in developing bispecific antibodies which have the capacity to go after two targets with one antibody. Amgen is paying $900 million upfront, though potential milestone payments could push the deal’s value up to $1.6 billion. TeneoBio is developing antibody technologies that include one therapy, currently in a Phase 1 clinical trial, aimed at treating certain forms of metastatic prostate cancer. The deal is expected to close in the second half of 2021. MTSL rec CLDX was up on the news in sympathy as they also have a bispecific antibody platform.
Sanofi is acquiring Translate Bio for $38 per share/$3.2 billion. The purchase price represents a premium of 56% to Translate Bio’s volume-weighted average price per share over the past 60 days. The company is a leader in the mRNA medicine space which restores protein function and is red hot after the recent success of the mRNA vaccines.
Bayer is buying Vividion Therapeutics, a private San Diego company that focuses on drug development using protein surface screening for $2 billion. Vividion only recently filed for an initial public offering (IPO), which it has decided to abandon with this acquisition. Under the terms of the agreement, Bayer is paying $1.5 billion upfront and potentially $500 million in various milestone payments.
Bayer believes that Vividion’s technology is the most advanced in the industry, and it has demonstrated its ability to identify drug candidates that can target challenging proteins. It is estimated that 90% of proteins linked to disease can’t be targeted by current therapies because of no known binding sites for drugs. Vividion’s technology platform uses chemoproteomics, chemistry, and biology to find drug targets in previously “undruggable” proteins.
Technicals – Vaccines & M&A Spur Another Rebound
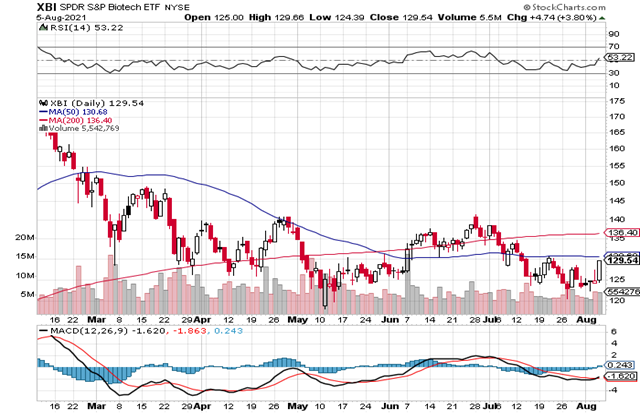
The wheel is turning and its can’t slow down. Right around press time, the XBI staged a very nice rebound based upon COVID-19 vaccine boosting, some merger and acquisitions activity, decent Q2:21 earnings by a few companies. A technical bounce off some recent index lows and possibly buying before the Fall rush also helped. The index closed near 130 and is now just below both the 50-day MA and the long-term 200-day MA (136), which does continue to creep higher. Fears of President Biden’s drug price reform have become more of a reality with the recent Executive Order but the large plans are still being bounced between Congress and many changes are likely before any final bills are enacted.The RSI has moved up nicely on the latest pop (to 53 from 36 three weeks ago) and is at neutral levels. The daily MACD is neutral again after coming off mid-July lows. So, there’s been some but not widespread bottom fishing among many names during the summer so far.
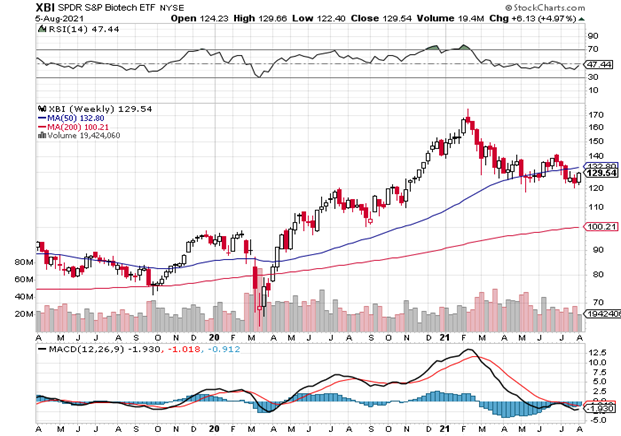
While the daily chart had a solid bounce, the long-term XBI (129.5) still is below support – notably the 50-week average. The weekly RSI (47) is near neutral but unlike last Issue, it is rising and not falling. The 50-week (133) keeps crawling slowly higher, but the index needs a solid move higher (e.g., 3-5% to break above that). There is still lot of room to fall to the 200-day (100) but with the increase in the vaccine leaders (MRNA, BNTX, NVAX), it feels like that won’t be happening anytime soon. The MACD had broken down a bit, but is now flattening. In biotech, things keep changing fast – and they just did again and again and yet again – and will probably once more. There is an increasing global demand for COVID vaccines and the Delta variant is not going away any time soon — and other future variants are possible.
Good News
Arcturus shares were up over 65% on news that the Singapore Health Sciences Authority (HSA) have approved the initiation of its mRNA COVID-19 vaccines ARCT-154 and ARCT-165. The company expects to initiate the trial in the coming weeks.
BeyondSpring was up over 170% after they announced that its Phase III DUBLIN-3 trial of plinabulin in combination with docetaxel to treat 2nd and 3rd line NSCLC (non small lung cancer) met the primary endpoint of increasing overall survival (mean OS, p = 0.03; HR=0.82). The trial also nailed the key secondary endpoints including that of progression free survival (PFS) (p<0.01), compared with docetaxel alone.
Bad News
Ardelyx was down over 75% after they announced the FDA has identified deficiencies with its New Drug Application for the control of serum phosphorus in adult patients with chronic kidney disease (CKD) on dialysis. The FDA specically noted the size of the treatment effect and its clinical relevance as one of the key issues.
Seres Therapeutics dropped over 60% after its Phase IIb ECO-RESET trial of SER-287 in patients with mild-to-moderate ulcerative colitis (UC), did not meet its primary endpoint of improving clinical remission rates compared to placebo.
Tonix Pharmaceuticals Holding Corp. was down over 35% after announcing that it will stop enrollment in its Phase III RALLY trial of TNX-102 SL for the management of fibromyalgia. The decision follows interim analysis by the Independent Data Monitoring Committee which recommended stopping the trial for futility.
Iterum Therapeutics was down over 30% after they announced that the FDA issued a Complete Response Letter (CRL) for its New Drug Application (NDA) for sulopenem. The FDA recommended that Iterum conduct at least one additional adequate and well-controlled clinical trial.
Annovis Bio released updated data from its trial of Posiphen in patients with Alzheimer’s and Parkinson’s disease. The data showed that patients on the placebo saw improved cognitive impairment compared with Posiphen. Despite the positive data at the Alzheimer’s Association International Conference (AAIC) the stock was down over 70%. Other stocks that presented data at AAIC also sold off on their presentations, with Cassava Sciences also down over 40%.
Clinical Trials Watch
Company Updates
ACAD — Nuplazid Revenue Misses, DPR Indication Still in FDA Limbo
ACAD recently reported Nuplazid sales revenue of $115.2 million compared to consensus estimates of $124.9 million for the quarter as the pandemic continues to impact a large part of the country. Although management noted new patient starts at par with the pre-pandemic level, they remarked that the starts have not accelerated as expected. Both in-person office visits and occupancy rates within the long-term care channel have receded as the pandemic continues to be disruptive to patient visits. Importantly, ACAD confirmed that it completed its Type A meeting with the FDA regarding the recent complete response letter (CRL) for Nuplazid in dementia-related psychosis.
ACAD noted that the FDA found ACAD’s presentation of new analyses (reviewing the clinical characteristics and features of psychosis among various dementia subtypes and demonstrating the similar clinical benefit observed across patients in the different subgroups) to be of interest and expressed a willingness to continue the discussion in a subsequent meeting. ACAD expects to meet with the agency later this year post compiling a briefing document (for a Type A, B, or C meeting), and management outlined three potential outcomes that could emerge post further discussion and additional analysis/review of the data.
- The FDA agrees that the available data are sufficient for approval of the broader DRP indication
- The FDA aligns that collected data for an individual indication (such as in a larger subtype such as Alzheimer’s disease psychosis) are sufficient, thus narrowing the scope of a label, but not requiring additional clinical work
- The FDA reiterates its request for additional clinical data on each of the individual subtypes in order to secure approval
Management noted that it is preparing for all three scenarios, and acknowledged that the agency has not discussed the specifics on what an additional trial would entail. Nonetheless, ACAD reiterated that Nuplazid’s existing patent life would support additional clinical work assuming the path forward is reasonable. We await further updates on the regulatory discussion and path forward after an additional agency meeting planned in 2H21.
ALKS — FDA Grants Fast Track to ALKS’ Nemavaleukin, Vivitrol Sales Picking Up
The FDA has granted Fast Track designation to ALKS’ nemvaleukin alfa (nemvaleukin), the company’s novel, investigational engineered interleukin-2 (IL-2) variant immunotherapy, for the treatment of mucosal melanoma. Earlier this year, the FDA also granted orphan drug designation to nemvaleukin for the treatment of mucosal melanoma. The company recently initiated enrollment in ARTISTRY-6, a global Phase II trial evaluating the anti-tumor activity, safety and tolerability of nemvaleukin monotherapy in patients with melanoma who have been previously treated with anti-PD-(L)1 therapy. The study is evaluating intravenously administered nemvaleukin in patients with mucosal melanoma and subcutaneously administered nemvaleukin in patients with advanced cutaneous melanoma. Nema has yet to be included in any Wall Street models and any good Phase II data will serve as a strong catalyst with fast track and orphan drug status both having been granted by the FDA.
Vivitrol sales were a surprise and have resumed to pre-COVID-19 levels. The primary Vivitrol driver has been the expanded use in alcohol dependent patinest along with better patient access and an improving net price. Looking ahead management expects Vivitrol alcohol dependence sales to increase in 2H21.
The company also believes that growing awareness of alcohol problems grew during COVID-19 and should drive longer-term segment growth. We are pleased to see ALKS maintaining some momentum with a solid earnings call followed by good news with Nema being granted FDA fast track status. Nema has yet to be included in any Wall Street models and any good Phase II data will serve as a strong catalyst with fast track and orphan drug status both having been granted by the FDA.
ESPR — ESPR Delivers Solid Nexletol/Nexlizet Revenue, Poised for Strong 2H21
ESPR reported 2Q21 earnings with US net product revenue coming in at $10.6 million (+67% QoQ) just below the consensus estimate of $10.9 million. As prescriptions grew 28% QoQ, management highlighted that adjustments have been made to patient assistance programs to help improve net pricing with limited impact on patient access. In our view, net pricing improvements will continue as well as positive trends for prescription volumes as the company shifts Nexletol/Nexlizet positioning and pushes formulary adoption further. The company also reported $1 million in royalty revenue from European commercial partner Daiichi Sankyo. We continue to look to the CLEAR outcomes trial as a huge factor for prescriber uptake. Speaking of CLEAR, ESPR stated that 75% of MACE events have thus far accumulated and the read out remains on track for 2H22. In our view, CLEAR will be positive and serve as a monster catalyst next year.
Rx demand during the quarter was up 28% from 1Q21, with >47,800 cumulative patients filling a script as of June 30. ESPR highlighted that the substantial improvement in net price in 2Q was driven by adjustments to both the duration and buy-down magnitude of the copay card program. Importantly, commercial and Medicare Part D coverage now stand at 95% and 60%, respectively, and the prescriber base is split roughly 60:40 primary care versus cardiologists.
Going forward, the company expects continued improvement in net price over time as plans fully implement coverage and volumes pick up. We expect ESPR to continue to build sales momentum in 2021 as both Nexletol/Nexlizet are very attractively priced for their substantial LDL lowering benefit. With much of Wall Street remaining skeptical, any positive sales growth in the 2H21 which results in a revenue beat will serve as a catalyst. ESPR remains under valued for a company with two approved drugs and a significant outcomes trial, CLEAR coming next year. In our view, CLEAR will be positive and serve as a monster catalyst next year.
MDGL — MDGL Prepares for Commercial Launch/ Leadership Team Expanded, NAFLD Phase III Up Next in Q321
MDGL is confidently preparing for the upcoming U.S. launch of resmetirom for both NAFLD and NASH. The company is building out its commercial resources and infrastructure, as well as developing a comprehensive product launch plan to position resmetirom for success. To date, MDGL’s market research projects have involved over 1,000 hepatologists, gastroenterologists and endocrinologists as well as payers who together provide prescription coverage for the vast majority of the patients in the U.S. Results from these efforts confirm that (i) NASH patients with significant liver fibrosis (F2/F3) are already being non-invasively identified by physicians, but given the lack of approved treatment options they are looking for effective new medications to manage the disease; and, (ii) the majority of these physicians believe an ideal therapy for NASH patients with significant fibrosis which addresses the underlying pathophysiology of the disease in the liver will slow, halt and/or reverse disease progression. In our view, MDGL’s management team is positioning resmetirom to become a billion dollar drug as the best in class treatment for both NAFLD and NASH.
Robert Waltermire is a key hire as the newly appointed Chief Pharmaceutical Development Officer of Madrigal within Research and Development with responsibility for all aspects of chemistry, manufacturing and controls (CMC) and commercial product supply. With over 30 years of experience in pharmaceutical development, Rob brings proven expertise in all aspects of chemistry, manufacturing and control (CMC) as well as experience in manufacturing new commercial products,” stated Becky Taub, M.D., President R&D and CMO of Madrigal.
Paul Friedman, CEO of Madrigal added, “We look forward to reconnecting with Rob at Madrigal after having worked with him at Dupont earlier in his career. Rob brings a wealth of experience to Madrigal, and helps us set the stage for commercial success.” Paul has always done an excellent job of reconnecting with his former collegues from Dupont where they developed Sustiva under Paul’s leadership as was the case when many of the Dupont chemists followed Paul to Incyte where he was previously CEO.
Next up is the pivotal Phase III NAFLD data in Q3. Positive data would be a significant stock catalyst and would set up an NDA submission for 2H22. Resmetirom could be FDA approved in 2023 which would position it as the only approved NASH drug for 2-3 years according to management’s estimates. We support this estimate as we have witnessed one competitor after another bite the dust. Management also highlighted recent market research confirming the large unmet need (~5mn people in the US with F2/F3 NASH, of which ~11% are diagnosed) and current lack of meaningful treatment options. In our view, MDGL’s management team is positioning resmetirom to become a billion dollar drug as the best in class treatment for both NAFLD and NASH.
PLXP — PLXP Delivers Three Partnerships With Walgreens, Rite Aid & CVS, Q2 Call Focus on Upcoming Launch
Our newest recommendation has hit the ground running with three new partnerships in the last three weeks as management executes flawlessly. First PLXP announced that three stock-keeping units (“SKUs”) of VAZALORE will be available in over 8,000 Walgreens stores nationwide later in August. Walgreens has already inserted placeholders to reserve space on shelves for all three SKUs: VAZALORE 81 mg, 12 count; VAZALORE 81 mg, 30 count; and VAZALORE 325 mg, 30 count. PLXP also announced that three stock-keeping units (“SKUs”) of VAZALORE, will be available in over 2,400 Rite Aid stores later in August. The third deal announced that three stock-keeping units (“SKUs”) of VAZALORE, will be available in nearly 8,000 CVS stores later this month. The retail price is expected to be $25 per month for VAZALORE.
On the company’s quarterly call we received more info on the imminent launch of VAZALORE which is coming this month.
Highlights of, and certain events subsequent to, the second quarter of 2021 include:
- Walmart, Walgreens, Rite Aid, and CVS among the leading retailers to reserve shelf space for all three SKUs of VAZALORE;
- Extensive distribution for VAZALORE in over 30,000 U.S. retail stores including Kroger’s and Target
- Launched specialty field force to raise awareness amongst healthcare professionals;
- Continue to inform specialists through medical conferences, publications and continuing professional education events; and
- VAZALORE launch inventory manufactured and currently shipping to retailers across the country.
Potential upcoming catalysts include the launch and the OTC advertising campaign with a strong online presence and Q3 revenues which will be booked as soon as drug is received at the retailer’s warehouse. We should also get some updates on the ibuprofen candidate or new additions to the pipeline throughout 2021. The path to market for ibuprofen and other approved drugs with the PLx Guard platform delivery system is much easier as they can use the 505 FDA abbreviated pathway for approval which is allowed when new drugs use the same active pharmaceutical ingredients. We expect the company to also announce the second pipeline candidate, possibly in conjunction when they announce the ibuprofen data. Many current drugs have both gastric issues and could benefit from the much better bioavailability provided by PLx Guard. The platform also provides the ability to significantly extend IP with new and approved formulations which can also be developed faster/cheaper utilizing the 505 FDA abbreviated pathway to approval.
VXRT — FDA’s Clears VXRT’s Phase II Trial For S-Wuhan Vaccine Candidate to Begin Shortly, Phase Ib Noravirus Vaccine Can Boost Previously Vaccinated Patients
VXRT has announced that the FDA has cleared their Investigational New Drug application for an S-only oral tablet SARS-CoV-2 vaccine candidate. The company intends to explore multiple S-only constructs in clinical trials alongside the S+N construct that has already completed its Phase I trial. This decision is based on preliminary data from a current Non-Human Primate study which indicates that the S-only vaccine produced much higher serum antibodies than the one expressing both S and N proteins. This contrasts with the Phase I results from the S+N vaccine candidate that showed remarkable T-cell responses and a mucosal antibody response, but not as strong serum antibody responses. The Phase II clinical trial with the S-Wuhan construct is expected to start shortly.
COVID-19 Vaccine Developments
While the nation’s death and illness tolls have fallen markedly since the beginning of the last quarter, new variants such as the Delta strain continue to worry national political and health leaders. Vaxart broadened its research into the various COVID-19 strains while continuing its development of an oral tablet vaccine. Among the most significant developments in the second quarter:
- The FDA cleared Vaxart to move to its next phase of COVID-19 testing with a study of its next generation S-1 construct.
- Vaxart is manufacturing the vaccine necessary to start the Phase 2 clinical study of its S-Wuhan construct and expects to begin this study shortly.
- A Non-Human Primate study of the S&N construct along with S-Wuhan and S-South Africa constructs showed optimal performance by the S-Wuhan construct and also cross reactivity against all variants tested. The decision was made to advance the S-Wuhan vaccine construct into Phase II development.
VXRT also announced that data came from their12-subject Phase Ib blinded “booster” study evaluating the ability of its norovirus vaccine to boost immunogenicity. Study participants were initially vaccinated with Vaxart‘s oral norovirus vaccine in late 2019 and were vaccinated again between February and April 2021. Serum IgG and IgA antibody responses were significantly elevated 29 days post-boost immunization, with no difference in titer between subjects that had received a prior oral norovirus vaccine and those who had not previously been vaccinated.
Importantly, all seven participants who had been previously immunized with the oral norovirus vaccine elicited a similar broad range of immune responses to norovirus as the five subjects that had not received a prior oral vaccine dose. The norovirus trial results also suggest that VXRT‘s oral vaccines may not be hampered by certain antibody response challenges that can occur with injectable viral vector-based vaccines. The antibody immune response can limit the effectiveness of booster/follow on vaccines as they generate immune responses against both the viral vector as well as the intended antigens.
The next catalyst for VXRT will be the start of Phase II trials for the S-Wuhan construct. The new clinical trial will allow VXRT to compare the S-only and S+N vaccine candidates and put them in a position to decide which approach offers the best way forward for our COVID-19 vaccine development program, particularly in the face of emerging variant strains. In our view, the preliminary data from a current Non-Human Primate study which indicates that the S-Wuhan construct produced much higher serum antibodies than the one expressing both S and N proteins bodes well for the S-only vaccine. The “booster” data from norovirus vaccine is also important as it shows that their oral COVID-19 vaccine candidates can be used repeatedly as boosters to extend protection as the pandemic drags on worldwide. VXRT has the potential to be both a major player and part of the long-term vaccine solution.
The Back Page
| Symbol | Company | Orig.Rec. | Current | Target | Recommendation |
|---|---|---|---|---|---|
| ACAD | Acadia | 33.79 | 19.25 | 45 | BUY under $28 |
| ALKS | Alkermes | 10.13 | 27.98 | 55 | BUY under $35 |
| BMRN | BioMarin | 12.68 | 77.93 | 150 | BUY under $100 |
| CLDX | Celldex | 10.50 | 47.70 | 47 | BUY under $70 |
| ESPR | Esperion | 24.42 | 15.00 | 70 | BUY under $35 |
| INCY | Incyte | 5.88 | 76.92 | 95 | BUY under $75 |
| IONS | Ionis | 7.63 | 39.38 | 65 | BUY under $50 |
| MDGL | Madrigal | 17.00 | 88.82 | 275 | BUY under $200 |
| MYOV | Myovant | 13.74 | 20.87 | 45 | BUY under $30 |
| NKTR | Nektar | 4.66 | 15.87 | 60 | BUY under $35 |
| PCRX | Pacira | 15.78 | 61.30 | 90 | BUY under $70 |
| PGEN | Precigen | 34.42 | 5.54 | 24 | BUY under $12 |
| PLXP* | PLx Pharma* | 16.70* | 18.44* | 35* | BUY under $22* |
| SGMO | Sangamo | 4.77 | 9.97 | 30 | BUY under $20 |
| VXRT | Vaxart | 8.00 | 8.69 | 30 | BUY under $15 |
| ZIOP | Ziopharm | 8.00 | 2.31 | 12 | BUY under $5 |
| ZYNE | Zynerba | 8.00 | 4.53 | 16 | BUY under $8 |
*New recommendation.
*Portfolio transactions: Purchased $270K of PLXP in the Model Portfolio and $270K PLXP in the Traders Portfolio using margin.
THE MODEL PORTFOLIO*
| COMPANY | SHARES OWNED | TOTAL COST | TODAY’S VALUE |
|---|---|---|---|
| Long Positions | |||
| Acadia | 5,000 | 156,557 | 96,250 |
| Alkermes | 4.000 | 88,690 | 111,920 |
| Celldex | 16,666 | 174,993 | 794,968 |
| Esperion | 3,491 | 105,316 | 52,365 |
| Incyte | 1,294 | 34,817 | 99,534 |
| Ionis | 3,250 | 49,123 | 127,985 |
| Madrigal | 3,292 | 69,980 | 292,395 |
| Myovant | 7,500 | 103,853 | 156,525 |
| Nektar | 6,500 | 63,277 | 103,155 |
| Pacira | 2,500 | 63,887 | 153,250 |
| PLx Pharma* | 16,167* | 269,988* | 298,119* |
| Precigen | 10,200 | 76,510 | 56,508 |
| Sangamo | 20,479 | 253,596 | 204,176 |
| Vaxart | 31,250 | 250,000 | 271,563 |
| Ziopharm | 27,500 | 166,100 | 63,525 |
| Zynerba | 10,730 | 150,003 | 48,607 |
| (08/05/21) | Equities: | $2,930,846 | |
| Cash: | $731 | ||
| PORTFOLIO VALUE: | $2,931,577 |
*The Model Portfolio is designed to reflect specific recommendations. We began the Model Portfolio on 12/23/83 with $100,000. On 4/13/84, we became fully invested. All profits are reinvested. Stocks recommended since then may be equally attractive, but may not be in the Model Portfolio. Transactions and positions are valued at closing prices. No dividends are created, and we don’t use margin. Interest income is credited only on large cash balances.
The Model Portfolio
THE TRADER’S PORTFOLIO**
| COMPANY | SHARES OWNED | TOTAL COST | TODAY’S VALUE |
|---|---|---|---|
| Long Positions | |||
| Acadia | 5,000 | 156,557 | 96,250 |
| Alkermes | 3,500 | 83,184 | 97,930 |
| Celldex | 16,666 | 174,993 | 794,968 |
| Esperion | 4,075 | 100,005 | 61,125 |
| Incyte | 2,229 | 51,176 | 171,455 |
| Ionis | 3,300 | 53,501 | 129,954 |
| Madrigal | 2,910 | 49,964 | 258,466 |
| Myovant | 7,410 | 102,831 | 154,647 |
| Nektar | 6,000 | 36,411 | 95,220 |
| Pacira | 2,000 | 55,918 | 122,600 |
| PLx Pharma* | 16,167* | 269,988* | 298,119* |
| Precigen | 10,170 | 119,952 | 56,342 |
| Sangamo | 20,479 | 253,596 | 204,176 |
| Vaxart | 31,250 | 250,000 | 271,563 |
| Ziopharm | 27,500 | 166,100 | 63,525 |
| Zynerba | 7,153 | 99,997 | 32,403 |
| (08/05/21) | Position Total: | $2,908,742 | |
| Margin: | -$1,281,890 | ||
| PORTFOLIO VALUE: | $1,626,852 |
**The Trader’s Portfolio joined the Model Portfolio on 1/6/05 with $500,000 and is designed to take advantage of short-term opportunities throughout the biotech sector. The Trader’s Portfolio will hold both long and short positions in stocks, trade-in options, and use margin. These strategies increase risk. Although there is no limit on the time any purchase can be held, the time frame for most investments will be weeks to months.
The Trader’s Portfolio
BENCHMARKS
| NASDAQ | S&P 500 | MODEL | TRADER‘S | |
|---|---|---|---|---|
| Last 3 Weeks | 2.4% | 1.6% | 5.1% |
9.1% |
| 2021 YTD | 15.5% | 17.9% | 10.0% | 19.3% |
| Calendar Year 2020 | 43.6% | 16.3% | 13.8% | 29.1% |
| Calendar Year 2019 | 35.2% | 28.8% | 10.7% | 44.1% |
| Calendar Year 2018 | 5.7% | 6.6% | 4.5% | 11.2% |
| Calendar Year 2017 | 29.3% | 19.9% | 65.6% | 98.9% |
| Calendar Year 2016 | 7.5% | 9.5% | -29.6% | -30.5% |
| Calendar Year 2015 | -0.1% | -0.1% | 25.1% | 27.9% |
| Calendar Year 2014 | 13.4% | 11.4% | 29.2% | 45.0% |
| Calendar Year 2013 | 38.3% | 29.6% | 103.4% | 214.7% |
BENCHMARKS
New Money Buys
NEW MONEY BUYS
Contact Info
Medical Technology Stock Letter
John McCamant, Editor
Jay Silverman, Editor
Jim McCamant, Editor-at-Large
Joan Wallner, Associate
BioInvest.com
PO Box 40460
Berkeley, CA 94704
510-843-1857
Send us an email
Download a PDF of MTSL Issue #957
©Piedmont Venture Group (2021). Address: P.O. Box 40460, Berkeley, CA 94706. Telephone: (510) 843-1857. Fax: (510) 843-0901. BioInvest.com. Email: admin@bioinvest.com. Published 24 times a year. Email subscription rates: 1 year – $399; 2 years – $678; 3 years – $898. You may cancel at any time for a prorated refund. The information and opinions contained herein have been compiled or arrived at from sources believed to be reliable but no representations or warranty, express or implied, is made as to the accuracy or completeness. In no way shall this newsletter be construed as an offer to sell or solicitation of an offer to buy any securities. The publisher and its associates, directors or employees may have positions in, and may from time to time make purchases or sales of, securities mentioned herein. We cannot guarantee and you should not assume that future recommendations will equal the performance of past recommendations or be profitable.