MTSL Issue 930
IN THIS ISSUE: Competitors’ CRLs Help MTSL Recs MDGL & PCRX
Since Last Issue: BTK: 3.3%; NBI: 3.7%; XBI: 6.7%; Model Portfolio: 4.8%; Trader’s Portfolio: 12.3%
SENTIMENT — Two CRLs Don’t Stop The Good Vibrations
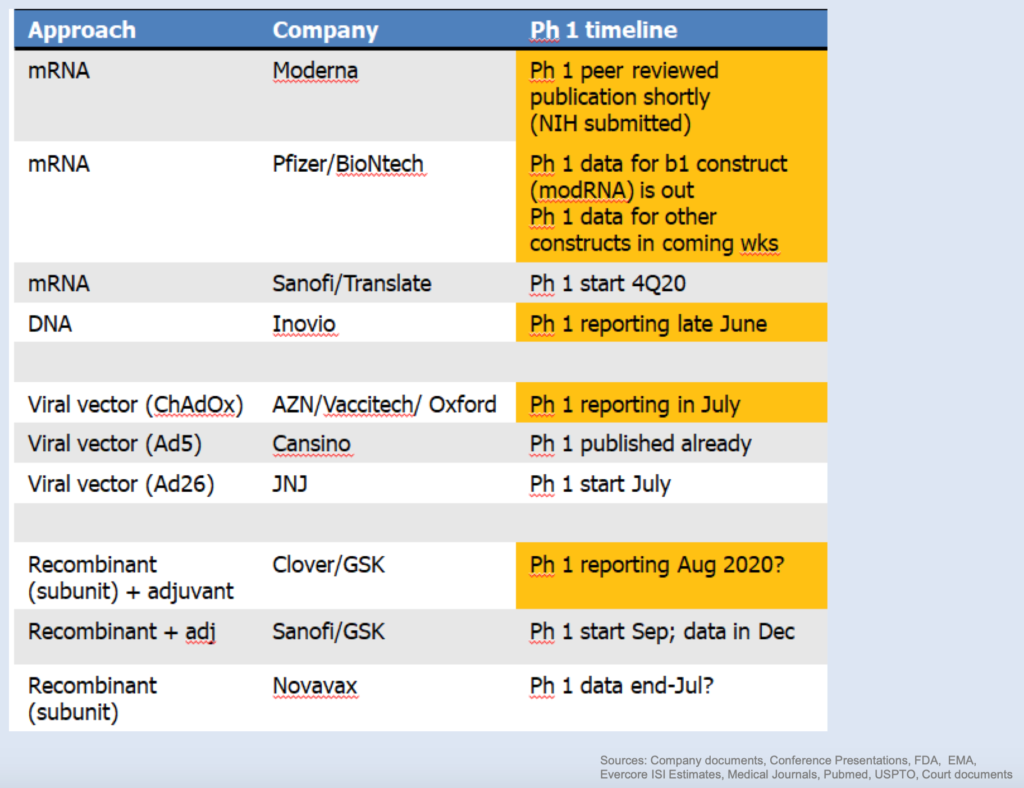
COVID-19 DRUG DEVELOPMENT UPDATE – FDA Issues Latest Vaccine Guidance, Countries Vie For Vaccines – There are now 2,378 global coronavirus-related studies posted on ClinicalTrials.gov (https://clinicaltrials.gov/ct2/results?cond=COVID-19). There are now more than 10 million people that have tested positive, many in the U.S., causing re-closures in several regions. Multiple vaccines are releasing initial data in July including MRNA, AZN/Oxford, PFE/BNTX, INO and NVAX. Remdesivir pricing announced. Countries and companies establishing vaccine manufacturing capacity before any data. Some cytokine storm studies approaching, too. Investor sentiment still solid despite new guidelines suggesting more stringent vaccine development requirements and possibly longer timelines, never-ending secondary offerings and hot IPOs.
FDA Vaccine Guidance Causes Some To Pause
On June 30, the FDA posted updated guidance on the requirements necessary to gain approval and eventual commercialization for any COVID vaccine (https://www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-fda-takes-action-help-facilitate-timely-development-safe-effective-covid). In sum, the agency reiterated that while there are various approaches (some proven, some novel) to address the pandemic, “Right now, neither the FDA nor the scientific community can predict how quickly data will be generated from vaccine clinical trials. Once data are generated, the agency is committed to thoroughly and expeditiously evaluating it all. But make no mistake: the FDA will only approve or make available a COVID-19 vaccine if we determine that it meets the high standards that people have come to expect of the agency.” Most important, the FDA tells us that traditional Phase III trials will be necessary and despite the urgency, there will be no shortcuts allowed. That and some uninspiring early data by INO, has paused the meteoric rise in the story stocks of today. Even for new leader BNTX (see below).
Remdesivir Pricing Below ICER Recommendation
Gilead announced pricing of its Covid-19 antiviral drug remdesivir. While the figure is lower than some estimates, shares of Gilead are bouncing on the news. A five-day course of remdesivir will cost private health insurers in the U.S. $3,120, and will cost governments of developed nations, including the U.S., $2,340. The Company believes that remdesivir’s ability to shorten recovery times for Covid-19 patients will result in hospital savings of $12,000 per patient in the U.S. It said it chose to set its price for remdesivir lower “to ensure broad and equitable access at a time of urgent global need.” The Institute for Clinical and Economic Review (ICER), an influential research group that issues drug pricing recommendations, said on June 24 that Gilead could charge between $4,580 and $5,080 per treatment course if remdesivir were priced based on the drug’s cost-effectiveness. Hence, pricing is lower than the ICER recommendation and in our opinion that should please providers and payors. Governments have already begun scooping up supply.
PFE/BNTX – Jumps To Lead
On July 1, a COVID-19 vaccine from PFE and BioNtech (BNTX) showed initial safety and produced strong neutralizing antibodies against the virus, keeping it at the forefront of the vaccine race. The messenger RNA product was tested on 45 healthy adults divided into several groups – 24 of them got two injections with two different doses of the experimental vaccine, 12 of them received a single shot with a very high dose and 9 patients got two placebo injections. All patients developed SARS-CoV-2 neutralizing antibodies 1.8- and 2.8-times the level of recovered patients. Low grade fever was a common adverse reaction (75%) in patients receiving the highest dose but only 8.3% in participants on the lower dose. A Phase 2b/3 trial is scheduled to start in late-July and will seek to confirm whether or not there is sufficient efficacy against preventing COVID-19. Additional data from three other candidates (BNT162a1, BNT162b2, BNT162c2) are expected over the coming weeks, after which they will select the best candidate to move into a larger trial involving as many as 30,000 patients. The study had been uploaded to the preprint server MedRxiv, but has yet to be peer-reviewed. Nonetheless, it appears to be solid enough to create the current leader in the race, while stocks of the competitors take a pause.
Inovio Early Data Does Not Say Much
INOVIO (INO) announced interim clinical data from its Phase 1 trial of INO-4800, its COVID-19 vaccine candidate. Limited data were released, with the company noting that 94% of trial participants demonstrated overall immunological response rates. However, key data regarding the number of patients that produced antibodies were not released. With the Company’s vague release, PFE/BNTX’s impressive data and the new FDA vaccine guidelines, INO shares have seen a sharp drop.
Vaxart (VXRT) Vaccine Pill Added To “Warp Speed” Project
Small cap biotech Vaxart’s oral COVID-19 vaccine candidate has been added to the cadre of experimental vaccines being studied as part of President Trump’s (along with DOD and HHS) mission to delivering 300 million vaccine doses against SARS-CoV-2 by January 2021. VXRT has a room temperature stable tablet vaccine that has just been selected for a non-human primate (NHP) challenge study organized and funded by Operation Warp Speed. The study is designed to demonstrate the efficacy of Vaxart’s COVID-19 vaccine candidate, which is based on the company’s Vector-Adjuvant-Antigen Standardized Technology (VAAST™) Platform. VXRT also announced a collaboration with Attwill Medical Solutions Sterilflow, LP (AMS) for lyophilization development and large scale manufacturing including tableting and enteric coating for Vaxart’s oral COVID-19 vaccine to manufacture as many as one billion doses a year. A lot can be said for Vaxart to gain acceptance in Operation Warp Speed, which so far includes the biggest and most advanced COVID vaccine players around.
SENTIMENT – Two CRLs Don’t Stop The Good Vibrations
Each of the past few times we have published the MTSL, the market’s Fear & Greed Index heads to overbought levels and then reversed back to neutral. The same pattern has occurred again with the F&G closing directly at 50, The XBI touched 70 (June 23) – which may have included some end-of-quarter window dressing – and now has receded to 62. Cities and states are recoiling from re-opening and the massive spike in infections is keeping businesses and government from moving to less restrictive phases – and that is probably good news for biotech stocks. Cash levels remain at record levels – and plush fund managers and funding biotech companies at a historic pace. After a sharp bounce when restaurants and bars started opening in June, underperforming sectors have quickly reversed. Biotechs are still kings of the castle in the current environment. After the flurry in May/June, the virtual science conferences are pausing. However, ZOOM presentations by companies themselves and sponsored by brokers are making it easier to reach a much larger audience from home. While we have been through a trove of positive data and deals and approvals, the FDA issued two Complete Response Letters (CRLs) that flat out rejected new drugs in the NASH and pain space, to ICPT and HRTX, respectively. MTSL Recommendations MDGL and PCRX are major beneficiaries of the delays. Despite the 40% and 33% respective declines in ICPT and HRTX, the CRLs failed to slow down the biotech stock party.
An FDA approval for RARE and favorable follow-on NASH data for ATRA highlight the good, non-COVID biotech industry news keep long investors in the right direction. With the giant joint ventures announced in the last Issue, SNY has not yet made any acquisitions with their REGN bounty. On the other hand, QURE’s stock fell after the Company signed a major deal with CDL Behring for its hep B gene therapy. QURE was rumored to be up for sale over the past year or two and failed to deliver a premium takeover. When SNY sold their REGN stake, some analysts predicted that Sanofi may buy QURE. The CSL Behring deal put an end to that speculation.
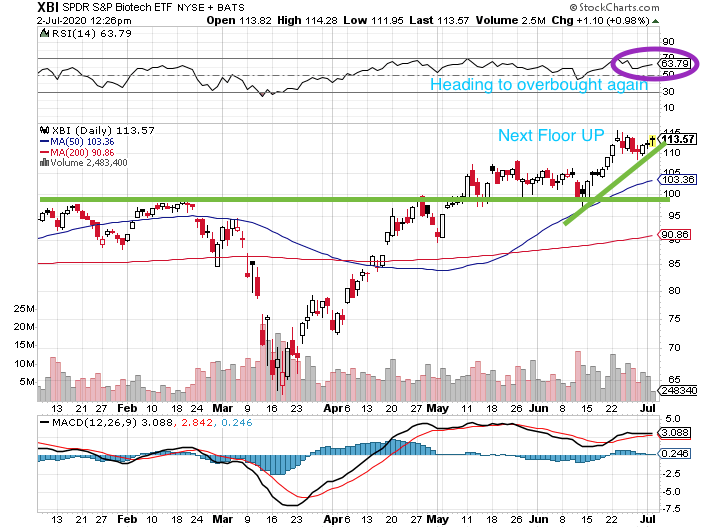
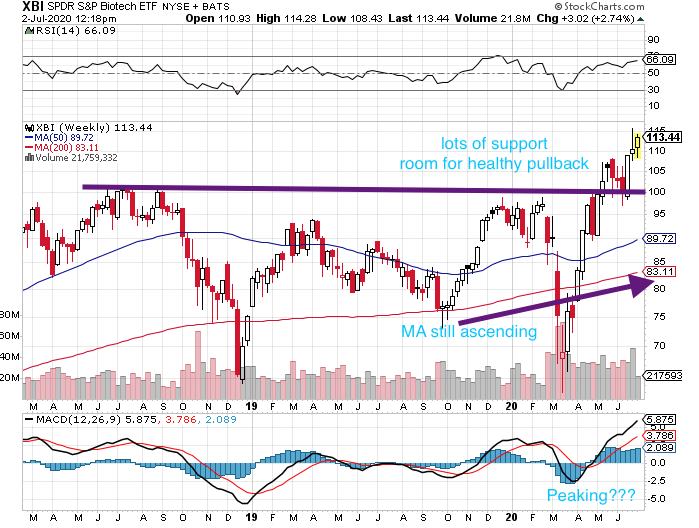
TECHNICALS – Next Floor Up Or Pie In The Sky
After basing for a month from mid-May to mid-June, the bios have moved another level higher as the powerful second quarter for the group came to an end. The XBI is currently at ~114, having bounced higher off the super strong support at 99 that created a wonderful technical floor. The next floor up came with the help of a fundamental catalyst from a large cap – notably MTSL Rec BMRN’s hem A data. While some stocks are at what we believe are “pie in the sky” valuations, the chart says we are still in good shape as a group (see XBI daily chart above). The index remains well above the rising 50-day (103) and 200-day (91) moving averages. The daily XBI RSI is at 64 (positive). The MACD is base lining a bit, probably due to the increased demand, huge cash inflows and some smart money profit taking. Not unexpected, the weekly XBI is nearing overbought (66) although the 50-week MA (90) and 200-week MA (83) keep rising. The weekly MACD (+5.9) is at bullish levels not seen in years. Hence, the charts say to us that biotech is getting a bit overheated, but still in a very strong position technically. There is a lot of room for a healthy pullback which should not deter the positive momentum.
FINANCING – The Gold Rush Continues
Big boys keep getting bigger – at least their balance sheets. While it is hard to add up all of the public and private fundraising occurring daily in Bioland, the amount is clearly an all-time record – probably approaching or exceeding $10 billion YTD. While there have been a few major takeovers of late, the largest development-stage biotechnology companies are adding Big Bio and Big Pharma type cash via these offerings. For example, SANA’s recent $700 million A round is an excellent example (SANA’s CEO is Steve Harr, who previously was CFO at Juno after running Morgan Stanley biotech banking. We worked with Harr back in our Robertson Stephens days in the late-90s). This cash hoard allows well-funded companies to compete at almost every level of drug development, and remain independent should they choose to do so. (We have always believed that SGMO is a prime takeover candidate due to the various Blue Chip partnerships they have in place, but management publicly has stated its opposition to being acquired at this stage. With ~$700 million in the bank and multiples of that in milestones and partner payments, that gives SGMO freedom to progress independently – despite some shareholders resentment.) Financial bounties are not only coming from investors, they are arriving by the truckload from global partners. The Biotech Gold Rush continues. The supply/demand equation will change someday, but it doesn’t look like that is happening any time soon.
This week’s select large financings:
- SANA $700 million (private)
- XLRN $500 million
- CRPT $500 million
- TBIO $275 million
- NLTX $75 million
DEALS – QURE/CSL Deal Says Takeovers May Be Less Common In A Flush World
After last Issue’s list of super corporate licensing deals, a like-sized deal was formed with gene therapy leader uniQURE and CSL Behring. CSL Behring will acquire exclusive global license rights to commercialize QURE’s AAV gene therapy program, AMT-061 (etranacogene dezaparvovec), for the treatment of hemophilia B. The AMT-061 program, currently in Phase III trials, could be one of the first gene therapies to provide potentially long-term benefits to patients with hemophilia B. Financially, uniQure may receive more than $2 billion, including $450 million in upfront cash, $1.6 billion in milestone payments, and royalties up to the low-20%.
While some smaller acquisitions are taking place, some behind the scenes, the current supportive financing market is allowing companies to raise huge money and remain independent. Stocks like QURE is just one example of a huge upfront payment and solid financial terms but remaining independent. Stocks of takeover speculative companies are therefore at risk should no full acquisition materialize. QURE had put itself up for sale but by the end of last year failed to find a buyer. The stock rallied (to 68) again on takeover rumors after an analyst said that SNY would buy them after selling its REGN stake. In addition, investors may have been valuing QURE for the much larger hemophilia A market, as BMRN stock has soared on its data and upcoming approval. Hemophilia B, however, is a much smaller market than hemophilia A. QURE shares closed at $46 at press time.
REGULATORY/DATA/APPROVAL – ICPT and HRTX Get CRLs – Good for MTSL MDGL & PCRX
The Intercept CRL for OCA was the biggest negative FDA surprise this year. We explain further in a MDGL update below, but it was one that most investors were optimistic might be the first approval for NASH. Subsequently, AKRO released further details on its previously announced positive NASH study with a 16-week analysis of secondary and exploratory endpoints in its Phase IIa BALANCED study of efruxifermin (EFX), formerly known as AKR-001. Notably, of the 40 treatment responders who had end-of-treatment biopsies, 48% achieved at least a one-stage improvement in fibrosis without worsening of NAFLD activity score (NAS) and 28% achieved at least a two-stage improvement in fibrosis. In addition, 48% of responders achieved NASH resolution with no worsening of fibrosis. Improvements in glycemic control and dyslipidemia, as well as weight loss, were also observed across all dose groups. Treatment with EFX was generally reported to be well-tolerated. AKRO leaps atop the NASH field, alongside MTSL Rec Madrigal’s resmetiron (see MDGL below).
Separately but on the same day, Heron (HRTX) received a second CRL for HTX-011, its liposomal bupivicaine for post-surgical pain. This one is a clear positive for another MTL Recommendation, Pacira (PCRX), of which we are raising our BUY LIMIT and TARGET PRICE on thanks to the HRTX CRL. See our PCRX update below.
RARE Gets Approval
For NMOSD – The FDA approved Ultragenyx’ (RARE) Uplizna (inebilizumab-cdon) injection for intravenous use for the treatment of neuromyelitis optica spectrum disorder (NMOSD) in adult patients with a particular antibody (patients who are anti-aquaporin-4 or AQP4 antibody positive). NMOSD is a rare autoimmune disease of the central nervous system that mainly affects the optic nerves and spinal cord. Uplizna is only the second approved treatment for the disorder. Hence, while two CRLs were unexpected, the FDA is still doing its job – granting approvals for drugs that have demonstrated safety and efficacy in the populations that they were tested in.
LEGAL – Drug Pricing Concerns Creep Its Ugly Head
While they might have liked to be given it for free, GILD’s remdesivir pricing is likely appeasing purchasers. Due to the virus, the high price of new drugs as a political target as well as any legislation concerning astronomical drug costs have been put aside by regulators for now. We have written before that, despite governments and the general public realizing the value of a good drug or vaccine more than ever before, eventually the political attacks will return. Most likely by the Presidential election this Fall. Rearing its ugly head, the Federal Court of Canada last week dismissed a plea by drugmakers challenging the Canadian government’s new regulations aimed at lowering prices of patented drugs. The court struck down one part of the new regulation, finding that using certain payments to third parties to determine a drug’s regulated price went beyond the government’s authority, but upheld the bulk of the rules. Overall investors disregarded the news (although drug stocks have taken a bit of a pause since).
Canada published the final regulations in August last year, despite strong opposition from drug companies. The lawsuit was filed in federal court and is led by Innovative Medicines Canada (IMC), which represents major pharmaceutical companies. The new rules will save money for patients, employers and insurers including the government at the expense of drug company profits. Here’s where is gets juicy – the Canadian reforms could spill into the drugmakers’ most lucrative market, the United States, where policymakers have suggested importing less expensive Canadian drugs or basing some prices on drug costs in other countries, including Canada. The plaintiffs included the Canadian units of PFE, ABBV, AMGN, BMY, AZN, NVS and LLY. The new regulations, which take into account cost-effectiveness of medicines and their impact on government budgets, apply only to drugs approved by Health Canada. The regulations have been deferred until January 1, 2021, from July 1. We do need to keep an eye on this.
AMGN Enbrel Patent Upheld
Unlike BIIB’s recent loss of a key Tecfidera patent that happened in the last Issue, this past week AMGN’s Enbrel patent was upheld by the courts. As a result, AMGN shares hit an all-time high. Patents are a critical value creator for new biopharmaceuticals (and why we like IP protected platform stocks such as IONS and SGMO with dominant patent estates).
Happy Summer!
Despite the beginning of summer vacations, biotech news flow is not expected to slow down. Less people are and cannot travel, creating added stress. Families are working from home and kids are enduring a really tough period in history. We wish everyone peace and happiness in this very difficult time and hope the novel coronavirus leaves and never returns. Following guidance for preventing the spread will only help that happen sooner than later.
Two major FDA submissions remain on the radar. MTSL Rec BMRN has a PDUFA date set for August 21 (we are optimistic), while the filing of BIIB’s Alzheimer’s drug is now due by late-2020. While this has certainly been an unprecedented period for biotechnology, MTSL remains astute in keeping our subscribers informed in a timely manner. Although we cannot obviously cover every company in the sector, we hope that our expanded Biotech Sector Analysis (BSA) at the beginning of each Issue gives readers the most important recent and upcoming industry events.
Despite the crazy and rather challenging world we are living in, we hope our readers have a wonderful, Happy & Healthy Fourth of July Weekend!
John & Jay & Connie & Joan
Clinical Trials Watch
Company Updates
ESPR — Phase III Data Published in the Journal of the American Medical Association (JAMA) Cardiology
ESPR just announced that pooled efficacy analysis from the four Phase III clinical studies of NEXLETOL, an oral, once-daily LDL-cholesterol lowering medicine, was published in the Journal of the American Medical Association (JAMA) Cardiology. The four Phase III clinical studies evaluated the efficacy and safety of NEXLETOL versus placebo in 3,623 patients with hypercholesterolemia while receiving stable lipid-lowering therapy and at high cardiovascular risk or with hypercholesterolemia and statin intolerance. JAMA is one of the world’s premiere medical journals and publication in this peer reviewed publication is a significant recognition.
The publication highlighted that NEXLETOL added to maximally tolerated statins, including moderate- or high- intensity or no background statin demonstrated significant additional LDL-C lowering levels versus placebo. In patients on background statin therapy, NEXLETOL lowered LDL-cholesterol (LDL-C) by a mean of 18% compared to placebo (p<0.001). In statin intolerant patients, NEXLETOL lowered LDL-C by a mean of 24% compared to placebo (p<0.001). Furthermore, decreases in non-HDL-C, total cholesterol, Apo B and hsCRP were greater with NEXLETOL versus placebo. Finally, overall and common adverse events occurred at similar rates in patients treated with NEXLETOL and placebo.
In our view, NEXLETOL is underappreciated by Wall Street as is the need and market opportunity to lower LDL in patients who are intolerant or underserved by statins. ESPR is a potential takeover target as it fits all the usual characteristics including a fully de-risked cardio pill that is FDA approved. Additionally, while the Company has signed very good partnerships for Europe and Japan, importantly ESPR has retained U.S. rights that make it very attractive as a takeover candidate. Any cardiovascular player (including those with PCSK-9 drugs) could add ESPR ($1.4 billion market cap) at a fraction of the December 2019 NVS $10 billion acquisition of MDCO – and without the regulatory/clinical risk or expense – including MRK, SNY, REGN, AMGN, PFE and/or Novartis, too.
MDGL — ICPT’s FDA NASH Setback A Positive For MDGL’s Resmetiron
ICPT recently received a CRL from the FDA for obeticholic acid (OCA) their potential treatment for NASH. Part of the problem was that ICPT appears to have expected the agency to focus on advanced fibrosis NASH patients. However, the FDA granted Breakthrough Therapy Designation (BTD) to ICPT for OCA in 2015 to treat NASH with fibrosis without specifying advanced fibrosis. By redefining a narrower target NASH population for OCA, ICPT may have created the problem with the FDA as the NDA was reviewed for a broader patient population. In our view, ICPT’s receipt of an CRL is a significant positive for MDGL as it narrows ICPT’s lead as they both race to become the first FDA approved treatments for NASH.
In addition, the focus on sicker NASH patients with advanced fibrosis allows for greater usage of non-invasive diagnostics, no biopsy, making it easier to penetrate this new market opportunity. The targeting of sicker NASH patients may support higher pricing for OCA. OCA is already approved as a second-line treatment for a unique liver disease, primary biliary cholangitis alone or in combination with ursodeoxycholic acid. ICPT sells OCA for almost $70,000 per year making it a very expensive drug. Even if ICPT were to sell OCA for $30,000 per year in the NASH market, they will have a difficult time competing with a cheaper and safer pill (i.e., resmetiron).
We expect resmetirom to provide clear efficacy benefits over OCA in NASH patients. The drug candidate will also have two distinct advantages – one of being a pill vs. an injectable; and two is huge price difference as MDGL has stated their intent to price resmetirom in the $5,000-6,000 range per year. In our view, resmetirom is the best-in-class NASH candidate with broad efficacy, the cleanest safety profile, NASH benefits, CV risk reduction, in an easy to deliver pill. All of these attributes will allow MDGL to aggressively target the broader NASH population, while OCA is limited to advanced fibrosis NASH patients.
MYOV — SPIRIT 1 Delivers Positive Data in Endometriosis, FDA Grants NDA Priority Review for Prostate Cancer – PDUFA Set For December 20th, Raising BUY to 23 and TARGET to 32
MYOV recently announced top-line results from SPIRIT 1, the second of two Phase 3 studies of once-daily relugolix combination therapy (relugolix 40 mg plus estradiol 1.0 mg and norethindrone acetate 0.5 mg) in women with pain associated with endometriosis. Relugolix combination therapy met its co-primary efficacy endpoints and all seven key secondary endpoints in the SPIRIT 1 study. In addition, relugolix combination therapy was generally well-tolerated and resulted in minimal bone mineral density loss over 24 weeks:
- Co-primary endpoints met with response rates of 74.5% for dysmenorrhea (menstrual pain) and 58.5% for non-menstrual pelvic pain (p-values < 0.0001)
- Women receiving relugolix combination therapy, on average, had a 73.3% reduction on the Numerical Rating Scale for dysmenorrhea from 7.3 (severe pain) to 1.8 (mild pain)
- Achieved all seven key secondary endpoints, including dyspareunia (painful intercourse) and a greater proportion of women not using opioids, with a generally well-tolerated safety profile including minimal bone mineral density loss
MYOV also announced that the NDA for relugolix (120 mg) for the treatment of men with advanced prostate cancer has been accepted for Priority Review by the FDA. The FDA grants Priority Review to applications for potential therapies that, if approved, would be significant improvements in the safety or effectiveness of the treatment, diagnosis, or prevention of serious conditions when compared to standard applications. The FDA has set a PDUFA date of December 20, 2020. In its acceptance letter, the FDA also stated that it is currently not planning to hold an Advisory Committee meeting for this application. If approved, relugolix would be the first and only oral gonadotropin-releasing hormone (GnRH) receptor antagonist treatment for men with advanced prostate cancer.
MYOV recently commented that the GnRH antagonists may have better cardiovascular profiles than GnRH agonists like leuprolide, and that HERO is the first prospective trial to demonstrate such advantage. Management has highlighted the 480% higher CV risk for leuprolide in the population with history of MACE and the 50% higher risk in the population without MACE history. Thanks to improved cancer treatments, cardiovascular mortality has now become the leading cause of death in patients with prostate cancer (~30% vs ~20% from prostate cancer alone without CV death). A CV benefit included in the clinical trials section of the label would be a huge win for MYOV and would serve as another significant marketing advantage for relugolix.
Data on the key secondary endpoint of PSA progression-free survival from HERO trial in prostate cancer in Q3:20 is the next upcoming catalyst for MYOV who has significantly delivered on all their 2020 catalysts this year. With five positive Phase III trials, Relugolix is now de-risked in three separate indications, uterine fibroids, endometriosis, and prostate cancer. The drug candidate has also been transformed into a “pipeline in a pill’ with three indications adding significantly to its net present value. We believe that the company is also a prime takeover candidate with a potential blockbuster drug about to hit the market. Taken together, we are raising our BUY to 23 (from 17) and our TARGET to 33 (from 25).
PCRX — HTX-001 Knocked Back Again, Lengthening Exparel’s Market Dominance
For at least the third and maybe the fourth delay (we’ve lost count), Heron Therapeutics (HRTX) received a second Complete Response Letter (CRL) from the FDA regarding HTX-011 for the management of postoperative pain. Once again, the FDA letter noted (four) non-clinical issues in the CRL. Three relate to confirming exposure of excipients in preclinical reproductive toxicology studies, and the fourth relates to changing the manufacturing release specification of the allowable level of an impurity based on animal toxicology coverage. While the HRTX bulls and PCRX bears will once more say there is no need for additional studies and the delay will be temporary, etc. etc. etc., Pacira’s Exparel leadership and monopoly for liposomal bupivacaine continues intact. It will likely be at least several months before HRTX can re-file and get approval, if ever.
Nonetheless, that company’s credibility with regards to HTX-011 continues to diminish while PCRX’s dominance continues to expand. In a world where the FDA appears to be bending over backwards to approve new drugs, another seemingly easy one keeps getting pushed back. This was an unexpected plus for PCRX, and with no real competition in sight again, we are raising our Buy LIMIT and TARGET PRICE.
PGEN – Presents Strong Preclinical Data for Ultra-CAR-T at AACR
PGEN recently announced preclinical data for its innovative investigational PRGN-3005 UltraCAR-T® in patients with advanced, recurrent platinum resistant ovarian, fallopian tube or primary peritoneal cancer has been published as an e-poster and accompanying audio presentation at the American Association for Cancer Research (AACR) Virtual Annual Meeting II. The e-poster presentation titled “PRGN-3005 UltraCAR-T: Multigenic CAR-T Cells Generated Using Non-viral Gene Delivery and Rapid Manufacturing Process for the Treatment of Ovarian Cancer” (Abstract 6593) was part of the Immunology/Adoptive Cell Therapy session and is accessible on the AACR e-poster website (June 22). Despite COVID-19, companies like PGEN are doing a good job of continuing data flow to the investment community via virtual conferences and posts online. In our view, management’s ability to start this trial during COVID-19 is a testament to both their skill and experience and illustrates a willing patient population for new cell therapies to treat cancer. Reiterate BUY.
PRGN-3005 is an autologous CAR-T treatment simultaneously expressing three gene products, which results in a uniform, homogenous CAR-T cell therapy: 1) CAR to specifically target the unshed portion of Mucin 16 (MUC16), which is overexpressed on over 80% of ovarian tumors with limited expression found in healthy tissues; 2) membrane-bound IL-15 (mbIL15) to provide improved UltraCAR-T persistence and maintenance of preferred stem cell like memory phenotype; and 3) a kill switch to eliminate the CAR-T cells, if needed.
Preclinical data demonstrate the specificity and efficacy of using the rapidly manufactured PRGN-3005 UltraCAR-T cells for the treatment of ovarian tumors. Specifically, a single administration of PRGN-3005 showed significantly superior expansion and preferred memory phenotype of UltraCAR-T in vivo and significantly superior efficacy compared to traditional CAR-T resulting in all PRGN-3005 treated mice becoming tumor-free. Furthermore, re-challenging these tumor-free mice three months later with ovarian tumors for a second time (to simulate tumor relapse) led to the elimination of tumor burden without additional PRGN-3005 UltraCAR-T treatment. These data demonstrate the potential of UltraCAR-T cells to persist long-term in vivo, prevent CAR-T cell exhaustion, and mount a durable anti-tumor response with the ability to continue to respond upon tumor re-challenge.
Based on these preclinical results, the FDA approved the IND application, and the first-in-human PRGN-3005 Phase I clinical trial for advanced ovarian cancer is currently under way (clinical trial identifier: NCT03907527). The PRGN-3005 UltraCAR-T Phase I clinical study is an open-label, dose escalation study to evaluate the safety and maximal tolerated dose of PRGN-3005 UltraCAR-T delivered by intraperitoneal infusion (IP) or intravenous infusion (IV). The study population includes patients with advanced stage (III/IV) recurrent ovarian, fallopian tube, and primary peritoneal cancer who are platinum-resistant and have progressed after receiving standard-of-care therapies or are not eligible to receive available therapies with known clinical benefit. We expect to see the first data from this potential game changing data by year-end. Because the first data will be in cancer patients it has the potential to serve as significant proof-of-concept (POC).
In our view, PGEN’s Ultra-CAR-T therapy has the opportunity to be a game changing technology as it can be used on site as their proprietary non-viral gene transfer process eliminates the need for ex vivo off site manufacturing, thus dramatically reducing wait times for patients from weeks to one day after gene transfer. First-generation CAR-T therapies cost as much as $500,000 per patient and PGEN should be able to price their CAR-T therapies at a much lower price enabling the technology to be used in a much broader patient population. The kill switch is also valuable as it allows for the therapy to be stopped if any serious side effects begin to occur. Importantly, the preclinical data demonstrates both a strong initial expansion of T-cells which eliminates current tumors, it also shows a strong memory T-cell effect when re-challenged with cancer. This is the hallmark of I/O where the therapy not only eliminates the current cancer, it also provides sustained immune surveillance allowing for an appropriate T-cell response when new cancer appears.
ZIOP — Completes Enrollment of Phase II Controlled IL-12/Libtayo Combo Trial
ZIOP announced that 36 subjects have been enrolled in the Phase II clinical trial evaluating Ad-RTS-hIL-12 with veledimex (Controlled IL-12) in combination with Regenron’s PD-1 inhibitor Libtayo (cemiplimab-rwlc) for the treatment of recurrent or progressive glioblastoma (rGBM) in adults. Subjects in this multi-center trial were enrolled from seven hospitals specializing in the treatment of brain cancers across the U.S. Any clinical progress enrolling new patients during COVID-19 is impressive and also points out the desire of rGBM patients for new therapies as they currently have no real treatment options.
The open-label, single-arm Phase II trial (NCT04006119) is designed to examine Controlled IL-12 in combination with cemiplimab in 36 patients with rGBM, with the primary endpoints being safety and efficacy. Patients with rGBM scheduled for resection, who have not been treated previously with immune checkpoint inhibitors, received Ad-RTS-hIL-12 intratumorally at the time of surgical resection plus 20 mg of veledimex, an oral activator of Ad-RTS-hIL-12, daily for 14 days. Patients will also receive cemiplimab intravenously (350 mg) every three weeks until documented progression or withdrawal from the study. Trial investigators may enroll additional patients currently in screening.
The rationale behind the combo and supported by data recently presented at ASCO is that Controlled IL-12 results in the sustained influx of activated T cells and upregulation of PD-1 expression. In our view, the combo has interesting potential and Regeneron would be an excellent partner, as the two firms currently have a supply agreement in place. Regeneron is late to the PD-1 market and would love to have a combo that might work in the 70% of cancers which are PD-1 negative and do not respond to anti-PD-1 therapy alone. Some data could be available by year end. We will look for more exact timing on the next quarterly call as the company should have a better idea of when now that enrollment is complete.
ZYNE — [Special Update] Zygel Topline Results Fall Short of Statistical Significance, 80% of Patients With FMR1 Gene Had Positive Affect
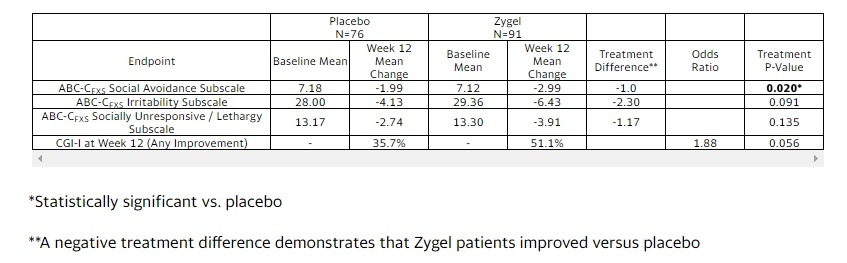
ZYNE’s stock is down significantly this morning after reporting disappointing top line results from the 14-week pivotal CONNECT-FX (Clinical study of Cannabidiol (CBD) in Children and Adolescents with Fragile X) trial. The multi-national, randomized, double-blind, placebo-controlled trial assessed the efficacy and safety of Zygel™ CBD gel as a treatment in for behavioral symptoms of Fragile X syndrome (FXS) in 212 patients failed to achieve statistical significance for the primary and secondary endpoints. Zygel did not achieve statistical significance versus placebo in the primary endpoint of improvement in the Social Avoidance subscale of the Aberrant Behavior Checklist – Community FXS (ABC-CFXS). Zygel also did not demonstrate statistical significance versus placebo in the three key secondary endpoints, which were the change from baseline to the end of the treatment period in the Irritability subscale score of the ABC-CFXS, the Socially Unresponsive/Lethargic subscale score of the ABC-CFXS and Improvement in Clinical Global Impression (CGI-I).
However, a pre-planned ad hoc analysis as defined by patients having full methylation of the impacted FMR1 gene showed an improvement of 40% vs 21.1% in 169 patients (p=0.002) in the primary endpoint, or 80% of the patients in the total trial. The Company believes that full methylation occurs in approximately 60% of the overall FXS patient population. Based on this analysis, Zynerba intends to meet with the FDA regarding a regulatory path forward for Zygel. While this is a subset analysis, it is not a small subset and represents more than 80% of the study (n=169/210), and based on the numbers alone it appears that the drug works in FXS patients with a fully methylated FMR1 gene as represented by the strong p=0.020. (i.e., p=0.05 is the level needed for statistical significance). In addition, the safety data was immaculate; with only 10% of subjects experiencing mild skin related irritation and no blood or liver side effects whatsoever.
Primary and Key Secondary Endpoints – Patients with Full Methylation of the FMR1 Gene
The results of CONNECT-FX in the analysis set of patients with full methylation of the FMR1 gene across the primary and key secondary endpoints are summarized below.
We are intrigued by the first biomarker for FXS patients as it may provide a path forward and potentially even allow the FDA to approve on this study. All that is required is a Southern blot/PCR test which is routine and relatively cheap to perform (at ~$350 each) to determine FMR1 methylation status. The big question going forward is will the FDA approve the subset with a biomarker. Given that this trial took 2 years from start to finish, 2 years would be a reasonable time frame to conduct another trial if the FDA requests one.
Upcoming Milestones:
- Developmental and epileptic encephalopathies (DEE): The results of discussions with FDA on the positive Phase 2 BELIEVE results and the clinical path forward are expected in Q3:20.
- Autism spectrum disorder (ASD): Zynerba intends to meet with FDA to discuss the positive Phase 2 BRIGHT trial results (released this May) and clinical path forward (H2:20 meeting).
- 22q11.2 deletion syndrome (22q): As a result of COVID-19 travel restrictions in Australia, top line Phase 2 data from the INSPIRE trial are now expected in Q4:20.
While we are disappointed, to us there does appear to a path forward for Zygel in FXS patients that have a fully methylated FMR1 gene – representing about 60% of the U.S. population (40,000/70,000). The big question going forward is will the FDA approve the subset with a biomarker. In our view, the addition of the biomarker may sway the FDA to approve on the subset as the agency is comfortable providing limited approvals that are scientifically defined. A best case scenario would be the agency granting conditional approval while ZYNE runs a confirmatory trial. With a market capitalization of $85 million with $60 million in the bank and several more catalysts and shots on goal, in our view, the downside from here is limited.
The Back Page
| Symbol | Company | Orig.Rec. | Current | Target | Recommendation |
|---|---|---|---|---|---|
| ACAD | Acadia | 33.79 | 51.61 | 60 | BUY under $46 |
| ALKS | Alkermes | 10.13 | 19.59 | 55 | BUY under $35 |
| BMRN | BioMarin | 12.68 | 126.78 | 150 | BUY under $115 |
| ESPR | Esperion | 24.42 | 50.96 | 100 | BUY under $75 |
| FPRX | Five Prime | 16.29 | 6.12 | 16 | BUY under $8 |
| INCY | Incyte | 5.88 | 106.47 | 115 | BUY under $90 |
| IONS | Ionis | 7.63 | 60.79 | 90 | BUY under $75 |
| MDGL | Madrigal | 17.00 | 111.58 | 275 | BUY under $200 |
| MYOV* | Myovant* | 13.74 | 20.03 | 33* | BUY under $23* |
| NKTR | Nektar | 4.66 | 23.52 | 60 | BUY under $35 |
| PCRX* | Pacira* | 15.78 | 52.37 | 70* | BUY under $50* |
| PGEN | Precigen | 34.42 | 4.82 | 24 | BUY under $12 |
| SGMO | Sangamo | 4.77 | 9.63 | 30 | BUY under $20 |
| ZIOP | Ziopharm | 8.00 | 3.24 | 12 | BUY under $5 |
| ZYNE | Zynerba | 8.00 | 3.50 | 27 | BUY under $18 |
*new recommendation
THE MODEL PORTFOLIO*
| COMPANY | SHARES OWNED | TOTAL COST | TODAY’S VALUE |
|---|---|---|---|
| Long Positions | |||
| Acadia | 5,000 | 156,557 | 258,050 |
| Alkermes | 4.000 | 88,690 | 78,360 |
| Esperion | 3,491 | 105,316 | 177,901 |
| Five Prime | 7,250 | 91,136 | 44,370 |
| Incyte | 1,294 | 34,817 | 137,772 |
| Ionis | 3,250 | 49,123 | 197,568 |
| Madrigal | 3,292 | 69,980 | 367,321 |
| Myovant | 7,500 | 103,853 | 150,225 |
| Nektar | 6,500 | 63,277 | 152,880 |
| Pacira | 2,500 | 63,887 | 130,925 |
| Precigen | 10,200 | 76,510 | 49,164 |
| Sangamo | 20,479 | 253,596 | 197,213 |
| Ziopharm | 27,500 | 166,100 | 89,100 |
| Zynerba | 27,500 | 150,003 | 37,555 |
|
(07/02/20) |
Equities: | $2,068,404 | |
| Cash: | $422,677 | ||
| PORTFOLIO | VALUE: | $2,491,081 |
*The Model Portfolio is designed to reflect specific recommendations. We began the Model Portfolio on 12/23/83 with $100,000. On 4/13/84, we became fully invested. All profits are reinvested. Stocks recommended since then may be equally attractive, but may not be in the Model Portfolio. Transactions and positions are valued at closing prices. No dividends are created, and we don’t use margin. Interest income is credited only on large cash balances.
The Model Portfolio
THE TRADER’S PORTFOLIO**
| COMPANY | SHARES OWNED | TOTAL COST | TODAY’S VALUE |
|---|---|---|---|
| Long Positions | |||
| Acadia | 5,000 | 156,557 | 258,050 |
| Alkermes | 3,500 | 83,184 | 68,565 |
| Esperion | 4,075 | 100,005 | 207,662 |
| Five Prime | 8,020 | 124,919 | 49,082 |
| Incyte | 2,229 | 51,176 | 237,322 |
| Ionis | 3,300 | 53,501 | 200,607 |
| Madrigal | 2,910 | 49,964 | 324,698 |
| Myovant | 7,410 | 102,831 | 148,422 |
| Nektar | 6,000 | 36,411 | 141,120 |
| Pacira | 2,000 | 55,918 | 104,740 |
| Precigen | 10,170 | 119,952 | 49,019 |
| Sangamo | 20,479 | 253,596 | 197,213 |
| Ziopharm | 27,500 | 166,100 | 89,100 |
| Zynerba | 27,500 | 166,100 | 25,036 |
|
(07/02/20) |
Position Total: | $2,100,636 | |
| Margin: | -$888,942 | ||
| PORTFOLIO | VALUE: | $1,211,694 |
**The Trader’s Portfolio joined the Model Portfolio on 1/6/05 with $500,000 and is designed to take advantage of short-term opportunities throughout the biotech sector. The Trader’s Portfolio will hold both long and short positions in stocks, trade-in options, and use margin. These strategies increase risk. Although there is no limit on the time any purchase can be held, the time frame for most investments will be weeks to months.
The Model Portfolio
BENCHMARKS
| NASDAQ | S&P 500 | MODEL | TRADER‘S | |
|---|---|---|---|---|
| Last 2 Weeks | 2.6% | 0.5% | 4.8% |
12.3% |
| 2020 YTD | 13.7% | -3.1% | 8.1% | 38.7% |
| Calendar Year 2019 | 35.2% | 28.8% | 10.7% | 44.1% |
| Calendar Year 2018 | 5.7% | 6.6% | 4.5% | 11.2% |
| Calendar Year 2017 | 29.3% | 19.9% | 65.6% | 98.9% |
| Calendar Year 2016 | 7.5% | 9.5% | -29.6% | -30.5% |
| Calendar Year 2015 | -0.1% | -0.1% | 25.1% | 27.9% |
| Calendar Year 2014 | 13.4% | 11.4% | 29.2% | 45.0% |
| Calendar Year 2013 | 38.3% | 29.6% | 103.4% | 214.7% |
BENCHMARKS
New Money Buys
NEW MONEY BUYS
Contact Info
Medical Technology Stock Letter
John McCamant, Editor
Jay Silverman, Editor
Jim McCamant, Editor-at-Large
Joan Wallner, Associate
BioInvest.com
PO Box 40460
Berkeley, CA 94704
510-843-1857
Send us an email
Download a PDF of MTSL Issue #930
©Piedmont Venture Group (2020). Address: P.O. Box 40460, Berkeley, CA 94706. Telephone: (510) 843-1857. Fax: (510) 843-0901. BioInvest.com. Email: admin@bioinvest.com. Published 24 times a year. Email subscription rates: 1 year – $399; 2 years – $678; 3 years – $898. You may cancel at any time for a prorated refund. The information and opinions contained herein have been compiled or arrived at from sources believed to be reliable but no representations or warranty, express or implied, is made as to the accuracy or completeness. In no way shall this newsletter be construed as an offer to sell or solicitation of an offer to buy any securities. The publisher and its associates, directors or employees may have positions in, and may from time to time make purchases or sales of, securities mentioned herein. We cannot guarantee and you should not assume that future recommendations will equal the performance of past recommendations or be profitable.