MTSL Issue 916
IN THIS ISSUE: MDCO/NVS & BOLD/Astellas – A Closer Look
Since Last Issue: BTK: 10.3%; NBI: 8.0%; XBI: 11.4%; Model Portfolio: 20.9%; Trader’s Portfolio: 54.5%
SENTIMENT — Where Do We Go From Here?
A perfect storm of sector positives has most biotech investors smiling for the first time since April. Sentiment is almost uniformly positive and that is something to be both happy for and concerned about. A flurry of Fall medical meetings has revived interest in new drugs for various illness – acute, chronic and rare – and the beginning of December has two major conferences taking place now that have big news to pay attention to. Further helping the
sector’s advance, the Alzheimer’s disease conference (CTAD) delivered enough data on BIIB’s aducanumab to keep the bulls in and the crowded shorts at bay for now. The ASH meeting is occurring this weekend. Abstracts were released in November and suggest several new trial updates will keep the good times treating blood diseases rolling (sound a little macabre?) There are scores of companies planning data releases during ASH that will also lead to added volatility in share prices. In our BioInvest Universe, we recommended ACAD into CTAD and SGMO into ASH. While not every stock is going up and there’s been a few big busts (notably sad SAGE depression data at press time), after A November to Remember, the biotech bull is back. For now.
A November To Remember — Major Positives Resulting In A Biotech Bull Market
| Date | Positive Event |
|---|---|
| November 1st | AMGN pays $2.7 billion for a 20.5% stake in BGNE (+41%) |
| November 8th | CELG/BMY – Reblozyl gains Fast Track FDA approval for beta thalassemia |
| November 13th | MRK acquired closely-held Calporta for up to $576 million |
| November 14th | AMRN (+28%) – unanimous FDA AdCom vote for Vascepa label expansion |
| November 16th | NVS – Adakveo gains FDA early approval as the first targeted therapy to prevent vaso-occlusive crises (VOCs) with sickle cell disease |
| November 17th | MDCO presented positive details of the ORION-10 Phase III trial of inclisiran at AHA |
| November 18th | KRTX (+307%) – positive results from Phase II trial of KarXT in schizophrenia patients with acute psychoses |
| November 19th | MYOV (+207%) – positive Phase III HERO trial of relugolix in advanced prostate cancer |
| November 19th | CRSP/VRTX (+27%) – positive interim data (n=2) for CTX-001 in beta-thal and sickle cell disease |
| November 20th | ALNY (+32%) – FDA approved Givlaari for the inherited disease acute hepatic porphyria; approval comes just 3 1/2 months after filing |
| November 24th | MDCO (+62%) acquired by NVS for ~$9.7 billion |
| November 25th | GBT (+29%) – Oxbryta granted Accelerated Approval by FDA for sickle cell anemia |
| November 25th | ICPT (+28%) – the FDA accepted the NDA for obeticholic acid (OCA) for NASH and granted Priority Review |
| December 2nd | BOLD (+106%) acquired by Astellas for ~$3 billion |
At BioInvest, we have analyzed biotechnology stocks for over 30 years and never before witnessed a month with so many explosive value creating events quite like November 2019. The biotech stocks listed above experienced exceptional gains based on many unexpected positive announcements. The gains listed in the parentheses are measured from the day before the favorable news until we went to press. Not included above are stocks that may or not have had news specific to their company but whose respective gains were either directly or indirectly aided by good data, FDA actions and/or takeovers and also saw their stocks skyrocket higher (e.g., ARWR, ESPR, DRNA, IONS, CLVS, MDGL, SGMO, RLMD, KOD, etc.). Many also participated in the myriad of medical and investor meetings that have occurred daily of late and still continued this week, like CTAD, ASH and the ISI HealthCONx Conference (see DATA below). As mentioned above, mid-cap SAGE blew up in an unanticipated failure of SAGE-217. To put the magnitude of the miss in perspective, SAGE’s market cap dropped from $7.8 billion to $3.1 billion that day (12/5), a painful reminder of the inherent risk in biotech stocks and drug development. That event also helped put a pause in the biotech rally.
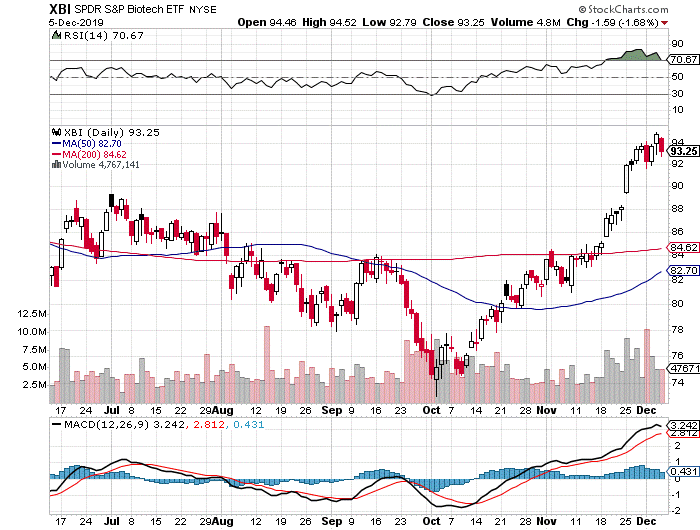
Technicals — What To Do After A Hockey Stick Move? The above list of sector catalysts, by no means exhaustive, has led to a massive rally in the IBB and XBI
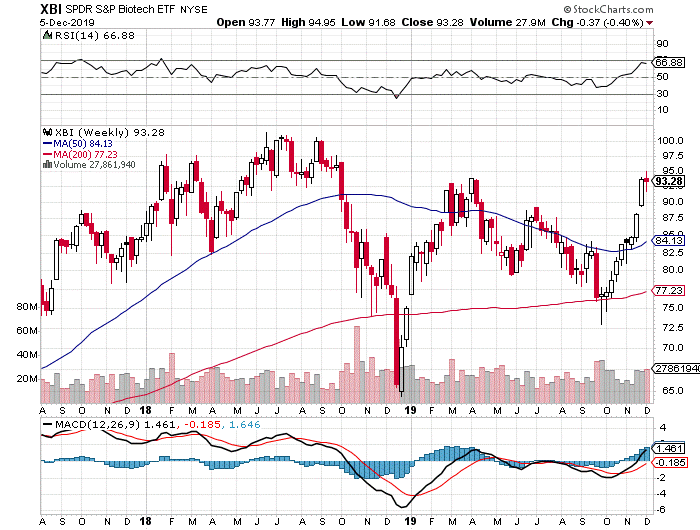
This Christmas holiday season has come early for biotech. The XBI has posted a positive weekly return for 9 of the last 11 weeks (with two flat weeks in the process but no negative ones). The 50-day (82) and 200-day (84) moving averages are way below as the XBI closed near 93, just a point below the high for 2019 that reached ~95 on Wednesday (94 was the level reached back in early April). For the past two weeks, the index was officially in overbought levels – it closed with an RSI of 71 at press time, but hit 83 on November 29. For several months, we believed that a breakout above the 200-day MA might lead to an explosive rally and with the abundance of good news that’s just what we got. The above plethora of good news – notably the surprise purchase of MTSL Recommendation MDCO by Novartis at an impressive premium only to be followed by the BOLD takeover by Astellas – led to a hockey stick move in the averages and even more impressive gains in individual names, before pausing on the SAGE disappointment.
The WEEKLY index, like the DAILY XBI, is way above support and looks a bit overextended (see XBI Weekly below). However, we also believe this is a strong representation of solid biotech fundamental health and investor optimism. With the daily RSI >70, some short-term pullback is inevitable. The daily MACD is strong but appears to have peaked near-term (+ 3.24). Like the daily index, it is also at the high for the year, and a short-term pullback is likely and would be considered healthy. The weekly MACD continues to look favorable but is also flattening a bit. The seasonal moves in stocks have tended to come earlier than usual this year – e.g., the summer rally happened in June and tax loss season occurred in September instead of October – one could presume that year-end mark-ups and window dressing are happening now and some profit-taking, therefore, may occur before year end. The smart money might be taking some off the table as we write. Most likely, however, is that fund managers, who had a difficult 2018, will try to milk 2019 for all its worth with the government in a stalemate and an impeachment looming for next year. High water marks and hurdle rates need to be recovered and then exceeded so the carried interest folks can buy whatever they want (except backbone). Speaking of fund managers’ greed, here’s a quick scene from one of our favorite movies, Bill Murray’s brilliant performance in Wes Anderson’s “Rushmore” (https://www.youtube.com/watch?v=m5RbdReBMLE).
M&A — A Little Closer Look At MDCO/NVS & BOLD/Astellas
In the previous Issue of MTSL, we said “M&A activity has been a bit quiet but as the year closes we believe M&A departments and bankers are working diligently to close some deals.” With the two big deals announced since then, we can take a lucky bow. Both deals were done at large premiums and at impressive valuations, but the two could not be more different in their respective strategic objectives. First, MDCO clearly was up for sale as it lacked the infrastructure to bring inclisiran to market in any meaningful way. The total ORION clinical package for LDL reduction was complete by the AHA meeting, and based upon the design of the CVOT trials compared with AMGN’s and REGN/SNY’s studies, we (and others) believe the odds of a greater reduction in CV risk including death are high. Partnering/takeover discussions were likely underway since at least the first successful Phase III was released, but once the additional trials were presented first hand to thousands of cardiologists, in our view, NVS’ bid was locked and loaded. Inclisiran’s value proposition was clear to us from the start, and Novartis and its cardiovascular franchise is a great fit (in addition to AMGN, REGN, SNY, PFE, etc.). In this case, the drug will likely gain FDA approval for LDL reduction by the end of next year, and label expansion over the next 2-3 years. We believe Inclisiran will be a blockbuster success and possibly a household name by then. Many MDCO bears did not believe a takeover was going to occur that quick, let alone at the ~$10 billion valuation. However, recent M&A transactions in biotech over the past 2-5 years suggested that Inclisiran was worth more than the market was discounting. The acquisition is a pure fundamental fit of a single blockbuster drug nearing commercialization. Most deals like this are focused on a single, lead compound although MDCO was unique in the sense that management bet the ranch and deservedly won. The MDCO/NVS deal gives us further confidence in the novel cardiovascular compounds and potential valuations of MTSL Recommendations ESPR and MDGL.
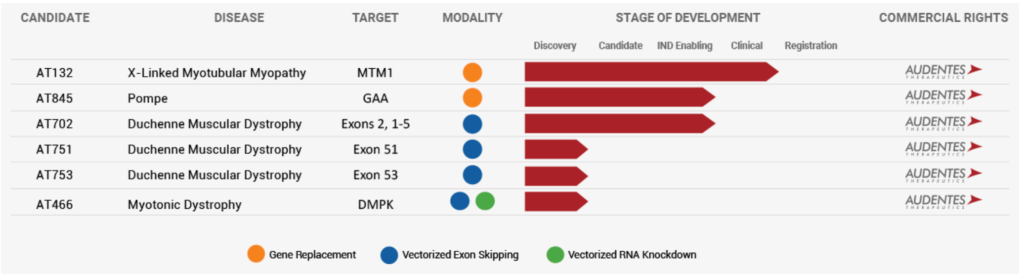
The Audentes (BOLD) acquisition by Japan’s Astellas is a platform deal that is focused primarily on AAV gene therapy. Gene therapy is one of the revolutionary technologies that is beginning to show real technological, clinical and even commercial success. The number of illnesses that may be corrected with gene therapy is formidable to say the least, and BOLD has a number of projects underway but only one in clinical trials, AT132, an AAV8 vector containing a functional copy of the MTM1 gene, for the treatment of XLMTM. Like many Orphan diseases, X-linked Myotubular Myopathy (XLMTM) is a rare, serious, life-threatening, neuromuscular disease that is characterized by extreme muscle weakness, respiratory failure and early death. Like many initial GTs, XLMTM is caused by mutations in a single gene, in this case the MTM1 gene that lead to a lack or dysfunction of myotubularin, a protein that is needed for normal development, maturation, and function of skeletal muscle cells. The disease affects approximately 1 in 40,000 to 50,000 newborn males. BOLD is expected to file for FDA approval of AT132 next year. As the table below shows, the rest of BOLD’s pipeline is in the pre-IND stage with a focus on DMD. Hence, we believe the deal represents Astella’s desire to gain access to novel future gene therapies and is paying $3 billion today to do so.
In our view, in every way this deal makes gene therapy/editing leader MTSL recommendation Sangamo look incredibly undervalued. When one takes into account SGMO’s breadth of patented technologies, long list of clinical trials underway and/or about to start, existing corporate partnerships and the remaining wholly-owned assets, we believe SGMO may be worth multiples that BOLD went for. In addition, MTSL has long recommended IONS, a pioneer in RNA targeted antisense technology that is also a revolutionary gene-related platform. IONS has three FDA approved drugs including one blockbuster in Spinraza, has multiple (>40) trials underway Ionis and its various collaborators). RNAI technology is also the source of MDCO’s Inclisiran (licensed from another RNAI and stock performance leader, ALNY).
REGULATORY – FDA On Speed – Early Nods For ALNY, NVS, GBT
At least four new drugs were approved by the FDA in November – all were okayed well ahead of their PDUFA dates. Usually a new drug is approved on or near the PDUFA and the recent spate of early regulatory authorizations is an excellent driver of stock performance. ALNY’s Givlaari (givosiran) is for the treatment of adult patients with acute hepatic porphyria, a genetic disorder resulting in the buildup of toxic porphyrin molecules which are formed during the production of heme (which helps bind oxygen in the blood). The PDUFA was February 4, 2020. Novartis. Adakveo (crizanlizumab) is a MAb approved to reduce the frequency of vaso-occlusive crises (VOCs) or pain crises in patients suffering sickle cell disease (SCD). The regulatory agency’s approval came two months ahead of the FDA’s priority review action date. Also in sickle cell disease, Global Blood Therapeutics (GBT) gained accelerated approval for Oxbryta (voxelotor), once daily tablets SCD. Both NVS and GBT approvals come right before the ASH meeting that focuses on blood disorders like SCD and beta thalassemia (CELG/BMY received Fast Track approval on Nov. 8th for their beta-thal drug, Reblozyl).
DATA – AUPH, BIIB, REGN Hit Big, CBAY, CARA & SAGE Miss Big
One autoimmune disease that’s been a tricky one for drug development is lupus. This week, two positive trials were announced – a Phase III study for AUPH’s voclosporin and BIIB’s Phase II LILAC study of BIIB059 showed benefits in patients with cutaneous lupus erythematosus (CLE) and systemic lupus erythematosus (SLE). (MTSL Recommendation NKTR has NKTR-358 in lupus trials with partner Eli Lilly.) Lupus has similar musculoskeletal symptoms as other autoimmune conditions such as multiple sclerosis. The AUPH data was another unexpected plus for biotechnology (as >9 million shares were sold short or 15% of the float as of 11/15/19), and its stock gained ~80% on the news.
There were a few big clinical flops since the last Issue. Two trials of CBAY’s lead candidate, seladelpar were halted mid-study due to liver damage side effects. The Company is terminating the NASH study as well as a trial with primary sclerosing cholangitis, an autoimmune liver disorder. (There is now one less competitor for MTSL Recommendation MDGL’s resmetirom.) Cara Therapeutics (CARA) announced disappointing results for its Korsuva drug which barely met significance in the highest of three doses and failed to meet a secondary endpoint in a study to treat patients with pruritis, a type of skin itching that comes from patients that have moderate or severe kidney disease. Before the negative news, both CBAY and CARA were relatively small, but overall well-liked stocks. Nonetheless, their bad news crushed both stocks, but has had a negligible effect on the biotech indices overall. The SAGE saga – a big one unfortunately – has led to pullbacks, profit-taking and probably some shorts to press their bets. After such a massive rally that included some huge short squeezes, SAGE gave investors a good reason to sell.
BIIB & ACAD At CTAD
This year’s Clinical Trials on Alzheimer’s Disease (CTAD) Annual Meeting was packed due to the presentation of clinical data for BIIB’s AZ Mab aducanumab, of which the Company is expected to file a BLA based upon the recent data and meetings with the FDA. AZ disease drugs have been one of the major black holes for big pharma and biotech, but the market is so potentially large that even a glimmer of efficacy could lead to sizable commercial sales. Hence, the CTAD data of the EMERGE and ENGAGE studies suggested minimal and difficult to analyze efficacy (i.e., data dredging) and a possibly acceptable but still not benign side effect profile (bleeding risks). The jury will be out on adumab until next year, and with the meeting behind us, the stock has recovered some after a pre-meeting bear raid as the data were not perceived as a complete bust.
MTSL Recommendation ACAD continued its remarkable 2019 comeback when the company presented further data from the HARMONY trial of pimavanserin in dementia-related psychosis at CTAD. As a reminder, the trial was stopped early in September due to positive efficacy, but the study details were not made public until this past week. More good news for biotech and the MTSL portfolio (see ACAD below).
ASH This Weekend For Hem A, Sickle Cell, Beta Thal, Blood Cancers
A good amount of news flow will come out from the conference – the last major one of the year. ASH has become as big as ASCO in many ways, and this year like the CTAD meeting, is one of the biggest ASH’s yet. There are so many data points to watch this weekend/next week including gene therapy for hemophilia A (BMRN, SGMO), initial gene editing data for CRSP (although previously disclosed) in sickle cell/beta thal (also for SGMO) and new multiple myeloma data from BLUE/CELG, AMGN, GSK and others. MTSL’s INCY will have a major presence with numerous abstracts, including data from its clinical development programs for ruxolitinib (Jakafi), itacitinib and ponatinib (Iclusig). So will now BMY-owned CELG. IL-15 preclinical data will also be presented. New CLL drugs are also on the docket. So while we may see some post-event sell-offs, in our view, ASH will give investors enough good news to ride 2019 out on its very high note.
FINANCE – Window Remains Wide Open
Some deals trade up, some down, but there are still lots of biotech/health tech secondary offerings and some IPOs priced on a daily basis. With the powerful rally in the past two months and despite the SAGE failure, more money is likely flowing into biotech funds, and managers will likely add to AUM into 2020 as well. Fund flows are probably the single most important variable in biotech sector stock performance, often driving the indices direction to overbought and oversold levels.
CALENDAR/KEY EVENTS – CTAD & ASH, Then Profit-Taking?
A few more big medical meeting remain in 2019, notably CTAD finishing this weekend and ASH beginning. After all of their clinical updates, SGMO will hold an investor day in NYC on December 17. Then the annual JPMorgan biotech shindig arrives before we know it (San Francisco, January 13-16). FDA filings for BMRN and MDCO are due by year-end.
- Phase III data for vosoritide (achondroplasia) – YE:19/Q1:20
- BLA filing for ValRox in the U.S. – by year-end
- Phase III GRAVITAS-301 trial for itacitinib in steroid naïve acute GVHD due by YE:19
- File Inclisiran BLA in the U.S. by YE:19
- SB-525 – hemophilia A Phase II update at ASH – December 7
- SB-920 STAAR – first patient enrollment in Fabry’s disease – YE:19
- ST-400 – additional preliminary data (Q4) in beta-thal at ASH – December 9
- TX-200 – initiate Phase I/II trial in solid organ transplant (RCC)
- Initiation of human trials for its novel TCR program – December?
- Phase 1 clinical trial of CD19-specific CAR-T, produced using a process termed rapid personalized manufacture (RPM) via Sleeping Beauty, for patients with relapsed CD19+ leukemias and lymphomas – before YE:19
Clinical Trials Watch
Company Updates
ACAD – Delivers Impressive Pimavanserin DRP Data at CTAD
ACADÂ reported details from its positive Phase III HARMONY trial in time to relapse of dementia-related psychosis (DRP) patients at the Clinical trials on Alzheimer’s Disease meeting (CTAD) and hosted a KOL panel.
Undeniable Efficacy
Pimavanserin nailed the primary endpoint with a significant 2.8x reduction in risk of relapse of psychosis versus placebo (HR=0.353; one-sided p=0.0023), stopping at the pre-planned interim analysis; and (2) met the key secondary endpoint with significant 2.2x reduction in risk of discontinuation for any reason (HR=0.452; one-sided p=0.0024). Based upon this, one can easily see why the HARMONY trial was stopped early.
Widening Therapeutic Window
On the KOL panel, the experts were impressed with the combined compelling therapeutic benefit with a significantly improved tolerability profile and no worsening of cognitive or motor symptoms. This was viewed as not just clinically meaningful, but also likely to reshape the treatment paradigm for DRP where there are no approved therapies. In our view, the data easily supports an sNDA filing in H2:20, following FDA discussions in H1:20.
Next Steps: Regulatory
ACAD plans to meet with the FDA in the first half of next year, an a supplementary NDA submission is probably coming in H2:20. At the end of Phase II meeting with the FDA, ACAD noted that a single well-controlled study with statistically and clinically relevant data could serve as the basis for the sNDA submission. The key was combining the efficacy data from the two previous acute studies and additional safety data from the ongoing placebo-controlled post-marketing commitment safety study of Nuplazid. ACAD is doing a very good job of creating a pipeline out of a drug with Nuplazid. As such, the stock is further de-risked increasing the chances of a premium takeover.
INCY – NDA for Pemigatinib Accepted By FDA
INCY recently announced that the FDA has accepted for Priority Review the Company’s NDA for pemigatinib, a selective fibroblast growth factor receptor (FGFR) inhibitor, as a treatment for patients with previously treated, locally advanced or metastatic cholangiocarcinoma with FGFR2 fusions or rearrangements. Importantly, Priority Review shortens the review period to 8 months compared to 12 months for Standard Review. The PDUFA target action date is May 30, 2020.
The NDA submission is based on data from the FIGHT-202 study evaluating pemigatinib as a treatment for patients with previously treated, locally advanced or metastatic cholangiocarcinoma. Study results, recently presented at ESMO demonstrated that in patients harboring FGFR2 fusions or rearrangements (Cohort A), pemigatinib monotherapy resulted in an overall response rate (ORR) of 36% (primary endpoint), and median duration of response (DOR) of 7.5 months (secondary endpoint) with a median follow-up of 15 months.
Pemigatinib is a potent, selective, oral inhibitor of FGFR isoforms 1, 2 and 3 which, in preclinical studies, has demonstrated selective pharmacologic activity against cancer cells with FGFR alterations. The drug development candidate is currently in multiple Phase II/III trials in various FGFR tumor types. Because FGFR is a growth factor which plays a key role in both tumor cell proliferation and survival, in our view, there is clearly potential for pemugatinib to become a cancer drug for multiple indications.
The next potential catalyst for INCY will be the Phase III GRAVITAS-301 trial for itacitinib in steroid naïve acute GVHD which is due before year end.  Given Jakafi’s positive outcome in steroid refractory acute GVHD, we expect the data to be positive despite the fact that steroid na¯ve patients require higher efficacy than refractory patients. With the recent M&A activity, we want to remind subscribers that INCY remains a prime takeout candidate with strong revenues and a wholly owned pipeline of oncology development assets.
MDCO – The Medicines Company (MDCO) – Novartis To Acquire The Medicines Company for $9.7 Billion or $85 Per Share (Special Update: 11/25/19)
From the beginning of the ORION clinical trials program in 2016 until the recent successful three Phase III studies, we have been under the belief that Inclisiran would be much bigger than people think. Today’s announcement tells us that Novartis, too, believe as such – as the consensus suggests that Inclisiran will need to generate at least $2.2 billion annually for the deal to be NPV neutral. In our view, the takeover will prove to be an astute acquisition at a very attractive price. The roughly $10 billion price tag is a reminder of the high value placed on innovation in biotechnology that is being recognized by global Blue Chip biopharmaceutical companies.
With a streamlined corporation structured solely on Inclisiran – we tip our hats to former CEO and MDCO founder Clive Meanwell for not only bringing the compound into the MDCO fold, but also designing the ORION clinical plan to perfection. Nothing in life is truly perfect, and the road from the test tube to treating patients and then to the market is filled with stops and starts. The Medicines Company has had its share of those over the years, but it certainly has come to a happy ending. Our previous price target on the day before the ORION-10 and -11 trial presentation was $85 and we raised it to $100 the day the stock jumped on takeover rumors. With both Boards of Directors having approved the merger, it strongly appears to be a done deal at $85 and hence, we do not expect any other bidders to emerge. Nonetheless, we and our MTSL subscribers are rather happy with the outcome. (https://www.novartis.com/news/media-releases/novartis-acquire-medicines-company-usd-97-bn-adding-inclisiran-potentially-transformational-investigational-cholesterol-lowering-therapy-address-leading-global)
Read Through To Other MTSL Names – ESPR, IONS, MDGL
The NVS/MDCO deal we believe has positive implications for other wholly-owned novel cholesterol drugs, such as Esperion’s BA and Madrigal’s resmetirom and we continue to recommend both stocks, currently trading well below their respective 52-week highs, aggressively at current levels. In addition, Inclisiran is an RNAI drug and the RNAI field continues to make steady progress of late clinically, with accelerated FDA approvals, reaching blockbuster sales levels in some cases and also developing next-generation technologies to improve safety and efficacy. As such, we also aggressively recommend IONS – an RNAI leader with over 40 compounds in the clinic and three approved drugs on the market – at current prices.
The MDCO takeover is yet another major positive for biotechnology stocks as they end 2019 with superb momentum.
- MDCO is a BUY under 70 with a TARGET PRICE of 100
- ESPR is a BUY under 75 with a TARGET PRICE of 100
- IONS is a BUY under 75 with a TARGET PRICE of 90
- MDGL is a BUY under 200 with a TARGET PRICE of 275
MDCO — Inclisiran Shines At AHA (Special Update: 11/19/19)
The oral presentations of the ORION-9 and -10 studies further confirm the revolutionary compound that is Inclisiran. On top of its durable LDL reductions and impeccable safety, the ease of use and high compliance rate, in our view, suggest blockbuster status is likely. With Phase III program successfully complete, we remain steadfast in expecting a much larger market for Inclisiran than the current stock price is forecasting. Raising BUY and TARGET PRICE.
ORION-10 results demonstrated LDL-C lowering of 58% at 17 months, comparable to PCSK9 antibodies with only 2 doses/year
In ORION-10, patients given Inclisiran resulted in placebo-adjusted LDL-C lowering of 58% (p<0.0001) at day 510 and time-averaged LDL-C reductions of 56% (p <0.0001) from days 90 through 540 compared to placebo. The reduction in LDL in ORION-10 was greater than the 54% seen in the first Phase III trial ORION-11. More important, treatment emergent adverse events (TEAS) were balanced across both arms exception for injection site reactions which were mostly mild and transient (inclisiran 2.6% vs placebo 0.9%). Mid-way through the trial, drug delivery was moved from a vial and syringe to a pre-filled syringe with a smaller needle gauge. Injection site pain/reactions dropped from 2.1% to 1.0%. Although not powered for statistics, in a pre-specified exploratory CV endpoint of a basket of unadjudicated terms relating to CV death, cardiac arrest, non-fatal MI and/or stroke, patients treated with inclisiran showed a marked decrease compared to placebo (7.4% for I vs 10.2% for P) similar to what was observed in ORION-11 (inclisiran 7.8% vs placebo 10.3%). A very small number of patients on Inclisiran experienced a CV death (n= Inclisiran 7 (0.9%) vs placebo 5 (0.6%), the numbers are insignificant and opposite although similar in ORION-11. While it is too early to determine the ultimate impact of inclisiran on CV outcomes the science and KOLs predict a significant reduction in MACE (e.g., conservatively 30% implied by Dr. Kastelein on the conference call).
ORION-10 & ORION-11 Pooled data showed remarkable risk ratio of 0.74 – or 26% reduction in CV risk – at 18 months
Because the trial designs and patient populations were very similar, in a presentation at AHA on Sunday night and again on today’s call, Dr. John Kastelein reported results from an ORION-10 & -11 pooled data analysis. In this combined dataset, inclisiran reduced LDL-C by 56% (p<0.00001) at day 510 (placebo-adjusted). Among the 1591 patients given inclisiran, 91% met a LDL-C threshold of <70 mg/dL and 87% demonstrated ≥50% LDL-C lowering – reaching AHA-recommended guideline goals. In the exploratory cardiovascular endpoint, inclisiran showed a risk ratio of 0.74 (95% CV: 0.59-0.93) representing a 26% risk reduction. Using the pooled data, MDCO projects a computed 5-year MACE relative risk reduction (RRR) of .70 for the currently enrolling, large ORION-4 CVOT study. As these estimates are based on LDL-C reductions observed in trials with likely adherence issues, Dr. Kastelein noted that while a 30% reduction in MACE is anticipated, he believes this number could be even higher due to the built-in adherence of inclisiran’s physician administered, twice yearly dosing regimen.
ORION-9, inclisiran delivered placebo-adjusted LDL-C reductions of 50% at 17 months in HeFH
This morning MDCO also announced that the ORION-9 trial demonstrated placebo-adjusted LDL-C lowering of 50% (p<0.0001) at day 510 and demonstrated time-averaged LDL-C reductions of 45% (p <0.0001) from days 90 through 540 compared to placebo – again on par with the LDL-C lowering range seen with PCSK9 antibodies (~40-60%). These improvements translate to an absolute reduction of 71mg/dL at day 510 and a time-averaged LDL-C reduction of 63 mg/dL. While a different (and smaller) patient population than ORION-10 and -11, Inclisiran continued to show a durable and potent LDL-C lowering paired with a clean safety profile in patients with HeFH – an inherited genetic form of hypercholesterolemia.
MDCO plans to submit both ORION-9 and ORION-10 data to peer-reviewed medical journals in the near future and reiterated guidance to file for inclisiran approval in the US in 4Q19 and in the EU in 1Q20.
CONCLUSION – Inclisiran Will Be Bigger Than People Think
Every day, 47,000 people die of cardiovascular disease and the global annual population in developing countries is over 40 million. There have been very few new cardiovascular drugs approved of late except for the PCSK-9 Mabs (Repatha and Praluent), There under-utilization and overall disappointing sales, although rising, we believe make Inclisiran consensus projections way too conservative. A great example, however, is PFE’s Lipitor. As the first- and best-in-class statin, Lipitor did just under $13 billion in sales in 2006 and 2007 (with a lot of competition). In our view, with it’s impressive profile and value proposition, and with the right marketing muscle and no near-term competition whatsoever, Inclisiran could easily eclipse Lipitor’s peak sales. The takeout potential of MDCO continues to rise and more-so with the final ORION-LDL data behind us. With that and a lower discount rate (now that the data is out and undeniably positive), we are raising our BUY LIMIT and TARGET PRICE.
MYOV — Time Loves a HERO as MYOV Delivers Excellent Phase III Data for Advanced Prostate Cancer (Special Update: 11/19/19)
Legendary blues rockers Little Feat song, “Time Loves a Hero,” says it all for MYOV today as their Phase III prostate cancer data from HERO trial (https://www.clinicaltrials.gov/ct2/show/NCT03085095?term=Myovant+HERO&draw=2&rank=1) was excellent. Patient MYOV investors were rewarded this morning as the Phase 3 HERO study of once-daily, oral relugolix (120 mg) met its primary efficacy endpoint and all six key secondary endpoints in men with advanced prostate cancer. These results support an NDA submission to FDA upcoming in Q2:20 and future regulatory submissions in Europe and Japan. Importantly, MYOV has retained all rights for the world’s two largest markets, the US and EU. Companies with proprietary assets that have de-risked Phase III data are significant M&A targets, and MYOV has joined other MTSL recommendations like MDCO and ESPR as a clear takeover candidate.
In the primary endpoint responder analysis, 96.7% (95% CI: 94.9%, 97.9%) of men receiving once-daily, oral relugolix achieved sustained testosterone suppression to castrate levels. A responder was defined as achieving and maintaining testosterone suppression to less than or equal to 50 ng/dL from Week 5 through Week 48. For the study to be successful, the lower bound of the 95% confidence interval of the response rate had to be at least 90%.
Five key secondary endpoints demonstrated superiority to leuprolide acetate, including rapid suppression of testosterone at Day 4 and Day 15, profound suppression of testosterone at Day 15, rapid suppression of prostate-specific antigen (PSA) at Day 15, and suppression of follicle-stimulating hormone (FSH) at Week 24 (all p-values < 0.0001). In addition, relugolix demonstrated non-inferiority to leuprolide acetate on sustained testosterone suppression through 48 weeks (96.7% vs. 88.8%, respectively) with a between-group difference of 7.9% (95% CI: 4.1%,11.8%), the primary endpoint required for regulatory submissions outside of the U.S. In addition, the pharmacodynamic results showed no testosterone flare after initiation of relugolix and mean testosterone levels returned to normal levels within 90 days after treatment discontinuation.
The overall incidence of adverse events in the relugolix and leuprolide acetate groups was comparable (92.9% vs. 93.5%, respectively). In the relugolix group, 3.5% of men discontinued the study early due to adverse events compared with 2.6% of men in the leuprolide acetate group. The most frequently reported adverse events, reported in at least 10% of men in the relugolix group, were hot flashes, fatigue, constipation, diarrhea, and arthralgia. Unadjudicated major adverse cardiovascular events were reported in 2.9% of men in the relugolix group versus 6.2% of men in the leuprolide acetate group. These events included non-fatal myocardial infarction, non-fatal stroke, and all-cause mortality. In this regard, relugolix is demonstrating a safety advantage which becomes more important as advanced PC patients are now living longer lives with the advent of new therapies, many of which are oral like Reguolix.
Prostate cancer is a very competitive space and the comparative CV safety of relugolix vs. Lupron was discussed by physicians on the call and they were quite enthusiastic about this safety finding. Notably, one of the two KOLs on the call mentioned that as care improves, including the use of more oral drugs, so does the life expectancy of prostate cancer patients, they anticipate that MACE outcomes and other safety measures will play an increasingly large role in choosing treatments. Additionally, it was mentioned that the potential for patients to rapidly come on and off of oral relugolix therapy makes intermittent treatment a new option. Research done by MYOV found a large majority of men with prostate cancer would prefer a pill to injections. In addition to safety, more convenient delivery represents another real advantage for relugolix compared to the depot competitors. In biotech, time certainly loves a hero and today MYOV is one.
SGMO — Data Updates Due For Hem A & Beta- Thal At ASH – BUY (Special Update: 12/2/19)
Sangamo will have a very busy December, with two clinical data updates coming at the American Society for Hematology (ASH, https://www.hematology.org/Annual-Meeting/). We remain steadfast that SGMO will emerge as the leader in gene therapy for hemophilia A with SB-525. Then the Company will follow that success with gene therapy for Fabry’s disease, treating its first patient before year end. Moreover, an update for SGMO’s ST-400 gene therapy for beta-thalassemia will lead the Company into investors’ beta-thal discussions. Lastly, SGMO will hold an Investor Day in NYC on December 17.
SB-525 – Hemophilia A Gene Therapy (with Pfizer)
Present follow-up patient data at ASH 2019
December 7 – “Updated Follow-Up of the Alta Study, a Phase 1/2, Open Label, Adaptive, Dose-Ranging Study to Assess the Safety and Tolerability of SB-525 Gene Therapy in Adult Patients with Severe Hemophilia “
Regulatory discussions underway for Phase III
ST-400 Beta-Thalassemia – Additional preliminary data at ASH 2019
December 9 – “Preliminary Results of a Phase 1/2 Clinical Study of Zinc Finger Nuclease- Mediated Editing of BCL11A in Autologous Hematopoietic Stem Cells for Transfusion-Dependent Beta Thalassemia”
Complete patient enrollment by year-end 2019
ST-920 – Gene Therapy for Fabry disease
First patient enrollment expected by year-end 2019
December 17 – Analyst/Investor Day, NYC
Based upon its own merits and the value the market is placing on CRSP’s recent gene editing release of two patients, we believe SGMO shares remain extremely undervalued. We would not be surprised if one day Sangamo ends up being owned by either Pfizer or Gilead.
ZIOP – Presents Strong GBM Update at SNO, Up Next ASH Abstract for IL-15 Gene Therapy
ZIOP presented new interim analyses from two ongoing sub-studies in its Controlled IL-12 platform, or Ad-RTS-hIL-12 plus veledimex (Ad+V), both as monotherapy and in combination with a PD-1 inhibitor, for the treatment of recurrent or progressive glioblastoma multiforme (rGBM) in adults, at the 2019 Society for Neuro-Oncology (SNO) Annual Meeting (https://www.soc-neuro-onc.org/SNO/2019Annual/2019AnnualHome.aspx).
“Survival of Subjects with Recurrent Glioblastoma Receiving Intra-tumoral Administration of IL-12 Managed with Low-dose Dexamethasone”
Interim Update Reported – key points highlighted in bold
- Decrease in tumor from baseline (time of Ad+V administration) resulting in a patient’s lesion being too small to measure, assessed as a partial response (per iRANO), with follow up ongoing
- Subjects were comparable to the Main study except a higher percentage in the Expansion substudy had multifocal disease vs. unifocal disease and fewer recurrences
- Based on study design there was a higher percentage of subjects in the Expansion substudy as compared with Main study (75% vs 40%) who received lowdose concurrent steroids
- Local, regulated IL-12 production using Ad+V in subjects with rGBM rapidly and safely activates the immune system • Peak serum IL-12 at Day 3 with downstream production of endogenous IFN-g peaking at Day 7 in Expansion substudy (and Main study)
- MRI findings of pseudoprogression were consistent with immune-mediated anti-tumor effects
- 20mg V subjects (Main + Expansion, n=20) with unifocal disease at entry, receiving low-dose steroids (defined as <20 mg cumulative dosing of dexamethasone) continued to show a trend towards longer median overall survival (mOS, 16.2 months)
- Adverse reactions (ARs) were consistent with prior studies of Ad+V, predictable, doserelated, and promptly reversible upon discontinuation of veledimex
- No drug-related deaths were reported
“PD-1 Inhibition can be Combined with IL-12 in Subjects with Recurrent Glioblastoma”
Interim Update Reported
- Decrease by 64% in a patient’s tumor from baseline (time of Ad+V administration) resulting in a partial response (per iRANO) with follow up ongoing
- Enrollment is complete per 3+3 (Ad+V and nivolumab) dose-escalation, as well as an additional 12 patients enrolled to the third cohort (highest dose)
- Mean follow up is 4.8 months (with a minimum of 0.9 months and a maximum of 16.9 months); active dosing is ongoing, and mOS has not been reached
- Serum IL-12 was detected in all subjects following initiation of Ad+V, which is consistent with previously reported data on Ad+V as monotherapy
- MRI findings of pseudoprogression, consistent with immune-mediated impact on tumor
- No dose limiting toxicities, no serious adverse events that were considered related to the combination with nivolumab and no clinically significant overlapping toxicities were observed
- Drug-related toxicities in the combination substudy were comparable to the Main study, being predictable, dose-related, and promptly reversible upon discontinuation of veledimex and with no drug related deaths
Summary: Novel Approach And Broad Application To Oncology
Ziopharm’s Controlled IL-12 platform (or Ad-RTS-hIL-12 plus veledimex (Ad+V)) places IL-12 within the tumor and drives T cells into the tumor. This enables the immune system to adapt leading to anti-tumor activity. IL-12 gene therapy can turn back on the immune system to tackle recurrent or resistant GBM that is often fatal. In addition, the ability to combine ZIOP’s IL-12 gene therapy safely with a PD-I inhibitor is very novel and represents a significant opportunity in a broad range of cancers. Next up for ZIOP is an abstract discussing their IL-15 gene therapy at ASH, which will be followed by the first patient treated in the highly anticipated Phase II trial using their TCR technology in patients with solid tumors.
The Back Page
| Symbol | Company | Orig.Rec. | Current | Target | Recommendation |
|---|---|---|---|---|---|
| ACAD | Acadia | 33.79 | 50.48 | 60 | BUY under $46 |
| ALKS | Alkermes | 10.13 | 20.65 | 55 | BUY under $35 |
| BMRN | BioMarin | 12.68 | 80.08 | 130 | BUY under $100 |
| ESPR | Esperion | 24.42 | 47.00 | 100 | BUY under $75 |
| FPRX | Five Prime | 16.29 | 3.77 | 16 | BUY under $8 |
| INCY | Incyte | 5.88 | 93.99 | 95 | BUY under $75 |
| XON | Intrexon | 34.42 | 5.36 | 24 | BUY under $12 |
| IONS | Ionis | 7.63 | 64.53 | 90 | BUY under $75 |
| MDGL | Madrigal | 17.00 | 114.20 | 275 | BUY under $200 |
| MDCO | Medicines Company | 31.98 | 83.90 | 85 | BUY under $60 |
| MYOV | Myovant | 13.74 | 16.78 | 25 | BUY under $17 |
| NKTR | Nektar | 4.66 | 19.86 | 60 | BUY under $35 |
| PCRX | Pacira | 15.78 | 45.35 | 55 | BUY under $40 |
| SGMO | Sangamo | 4.77 | 11.60 | 30 | BUY under $20 |
| ZIOP | Ziopharm | 8.00 | 5.09 | 12 | BUY under $5 |
| ZYNE | Zynerba | 8.00 | 5.65 | 27 | BUY under $18 |
| CRSP | Crispr | 58.39 | 70.71 | 40 | HOLD |
| EDIT | Editas | 36.13 | 30.39 | 26 | HOLD |
| NTLA | Intellia | 31.63 | 17.23 | 21 | HOLD |
*new recommendation
Portfolio Transactions: We will sell the MDCO positions in both the Model and Trader’s portfolios. At the moment we will sit on the profits and wait for new opportunities. Be on the lookout for Special Updates during the Holidays as we may find a buying opportunity between now and year end. We want to thank the MDCO management team and employees for creating so much value for their shareholders. And lastly a huge shout out to Clive Meanwell who basically stole the drug from ANLY and put the drug on a clear path to the market.
THE MODEL PORTFOLIO*
| COMPANY | SHARES OWNED | TOTAL COST | TODAY’S VALUE |
|---|---|---|---|
| Long Positions | |||
| Acadia | 5,000 | 156,557 | 252,400 |
| Alkermes | 4.000 | 88,690 | 82,600 |
| Esperion | 3,491 | 105,316 | 164,077 |
| Five Prime | 7,250 | 91,136 | 27,333 |
| Incyte | 1,294 | 34,817 | 121,623 |
| Intrexon | 10,200 | 76,510 | 54,672 |
| Ionis | 3,250 | 49,123 | 209,723 |
| Madrigal | 3,292 | 69,980 | 375,946 |
| Medicines Co | 4,600 | 77,400 | 385,940 |
| Myovant | 7,500 | 103,853 | 125,850 |
| Nektar | 6,500 | 63,277 | 129,090 |
| Pacira | 2,500 | 63,887 | 113,375 |
| Sangamo | 20,479 | 253,596 | 237,454 |
| Ziopharm | 27,500 | 166,100 | 139,975 |
| Zynerba | 27,500 | 150,003 | 60,625 |
|
(12/06/19) |
Equities: | $2,480,682 | |
| Cash: | $31,953 | ||
| PORTFOLIO | VALUE: | $2,512,635 |
*The Model Portfolio is designed to reflect specific recommendations. We began the Model Portfolio on 12/23/83 with $100,000. On 4/13/84, we became fully invested. All profits are reinvested. Stocks recommended since then may be equally attractive, but may not be in the Model Portfolio. Transactions and positions are valued at closing prices. No dividends are created, and we don’t use margin. Interest income is credited only on large cash balances.
The Model Portfolio
THE TRADER’S PORTFOLIO**
| COMPANY | SHARES OWNED | TOTAL COST | TODAY’S VALUE |
|---|---|---|---|
| Long Positions | |||
| Acadia | 5,000 | 156,557 | 252,400 |
| Alkermes | 3,500 | 83,184 | 72,275 |
| Esperion | 4,075 | 100,005 | 191,525 |
| Five Prime | 8,020 | 124,919 | 30,235 |
| Incyte | 2,229 | 51,176 | 209,504 |
| Intrexon | 10,170 | 119,952 | 54,511 |
| Ionis | 3,300 | 53,501 | 212,949 |
| Madrigal | 2,910 | 49,964 | 332,322 |
| Medicines Co | 4,250 | 127,405 | 356,575 |
| Myovant | 7,410 | 102,831 | 124,340 |
| Nektar | 6,000 | 36,411 | 119,160 |
| Pacira | 2,000 | 55,918 | 90,700 |
| Sangamo | 20,479 | 253,596 | 237,454 |
| Ziopharm | 27,500 | 166,100 | 139,975 |
| Zynerba | 27,500 | 166,100 | 40,414 |
|
(12/06/19) |
Position Total: | $2,464,340 | |
| Margin: | –$1,249,937 | ||
| PORTFOLIO | VALUE: | $1,214,403 |
**The Trader’s Portfolio joined the Model Portfolio on 1/6/05 with $500,000 and is designed to take advantage of short-term opportunities throughout the biotech sector. The Trader’s Portfolio will hold both long and short positions in stocks, trade-in options, and use margin. These strategies increase risk. Although there is no limit on the time any purchase can be held, the time frame for most investments will be weeks to months.
The Model Portfolio
BENCHMARKS
| NASDAQ | S&P 500 | MODEL | TRADER‘S | |
|---|---|---|---|---|
| Last 3 Weeks | 1.0% | 0.7% | 20.9% |
54.5% |
| 2019 YTD | 29.0% | 24.3% | 16.8% | 56.1% |
| Calendar Year 2018 | 5.7% | 6.6% | 4.5% | 11.2% |
| Calendar Year 2017 | 29.3% | 19.9% | 65.6% | 98.9% |
| Calendar Year 2016 | 7.5% | 9.5% | -29.6% | -30.5% |
| Calendar Year 2015 | -0.1% | -0.1% | 25.1% | 27.9% |
| Calendar Year 2014 | 13.4% | 11.4% | 29.2% | 45.0% |
| Calendar Year 2013 | 38.3% | 29.6% | 103.4% | 214.7% |
| Calendar Year 2012 | 13.4% | 15.9% | 25.7% | 68.7% |
BENCHMARKS
New Money Buys
NEW MONEY BUYS
Contact Info
Medical Technology Stock Letter
John McCamant, Editor
Jay Silverman, Editor
Jim McCamant, Editor-at-Large
Dave Wexler PhD., Editor
Joan Wallner, Associate
BioInvest.com
PO Box 40460
Berkeley, CA 94704
510-843-1857
Send us an email
Download a PDF of MTSL Issue #916
©Piedmont Venture Group (2019). Address: P.O. Box 40460, Berkeley, CA 94706. Telephone: (510) 843-1857. Fax: (510) 843-0901. BioInvest.com. Email: admin@bioinvest.com. Published 24 times a year. Email subscription rates: 1 year – $399; 2 years – $678; 3 years – $898. You may cancel at any time for a prorated refund. The information and opinions contained herein have been compiled or arrived at from sources believed to be reliable but no representations or warranty, express or implied, is made as to the accuracy or completeness. In no way shall this newsletter be construed as an offer to sell or solicitation of an offer to buy any securities. The publisher and its associates, directors or employees may have positions in, and may from time to time make purchases or sales of, securities mentioned herein. We cannot guarantee and you should not assume that future recommendations will equal the performance of past recommendations or be profitable.