MTSL Issue 1071
IN THIS ISSUE: CLDX The Ideal M&A Target
Since Last Issue: BTK: -3.3%; NBI: -2.4%; XBI: -1.3%; Model Portfolio: 0.4%
SENTIMENT — Rates/Inflation Keep Bios Under Pressure
Novo Teams Up With Hims & Hers
In the last Issue, we commented that Novo needs to make moves to address their arch competitor, Lilly, who has been taking market share from Novo in the GLP-1 market. Novo is teaming up with Telehealth company Hims & Hers to sell its GLP-1 drugs and stop selling the “copycat” compounded versions it has long advertised despite lacking IP. The move comes after Novo recently filed a lawsuit against Hims & Hers for selling “unapproved” knock-off versions of Wegovy and Ozempic. By aggressively suing H&H Novo was probably able to negotiate a deal with better terms. The partnership also make sense for Novo as Lilly has been killing them with their LillyDirect program which has been capturing and maintaining patients before Novo can get to them. The collaboration between the two companies will offer both the injectable versions of semaglutide and the recently approved oral version of Wegovy.
This shift in business models for H&H is prudent as federal regulators had started to crack down on the sale of compounded GLP-1s. The Department of Health and Human Services had referred Hims & Hers to the Justice Department for possible criminal violations, and last month, the FDA sent out warning letters to 30 telehealth companies for the “illegal” marketing of compounded drugs. In our view, Novo remains an aggressive shopper for new obesity assets like VKTX, WVE or SKYE and any new drugs can be added to their sales partnership with H&H if they want.
GPCR recently announced additional topline data from its Phase 2b ACCESS-II study for aleniglipron (oral GLP-1) in the treatment of obesity, along with other updated data looks, including further data from the ACCESS-I open-label extension (OLE), and the Company’s Body Composition study. Perhaps most surprising from the release is the observation of deeper weight loss with no plateau through week 44 in ACCESS-II and week 56 in the ACCESS-I OLE. Specifically, patients receiving aleniglipron showed an additional ~1-2% weight-loss from week 36 to week 44, to reach a total placebo-adjusted weight-loss of ~16% in the ACCESS-II extension at the 180/240mg dose levels. This trend was consistent with further ACCESS-I OLE data, where patients showed another ~2% weight loss from week 44-56. In our view, VKTX offers a better value than GPCR with more assets, ‘2735 oral and sub-Q, plus the amylin program, and the ‘2735 program has the potential to best in class with a superior safety profile.
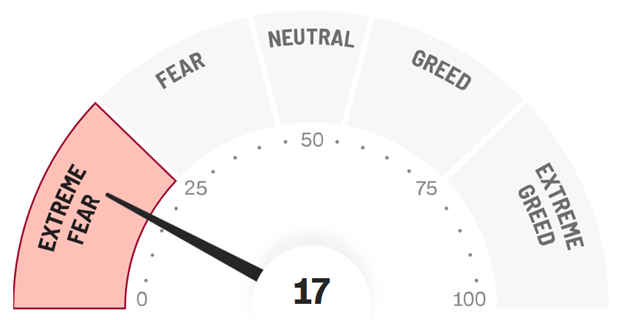
FEAR & GREED
The Fear and Greed Index has fallen into the dreaded Extreme Fear zone as the Iran war is causing significant economic uncertainty and the rising price of oil threatens to drive up inflation. Meanwhile, the Fed has voted to hold interest rates steady again in the range of 3.5%-3.75%, where it has stood since December. While Trump continues to pressure the Fed to lower interest rates, policy makers are acting with caution as they balance rising prices versus mixed signals from the job market. The longer the war lasts the less likely we are to see a cut as rising oil prices can be very inflationary.
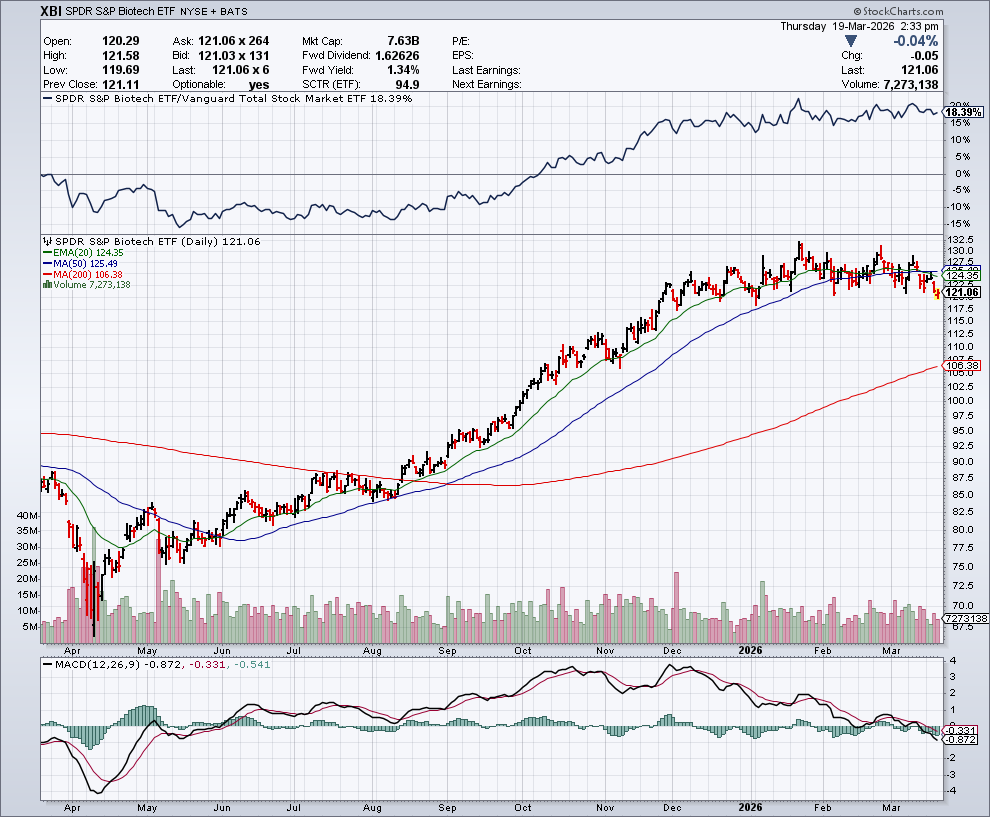
TECHNICALS – Rates/Inflation Keep Bios Under Pressure/XBI Below 50-Day MA
XBI Relatively Strong Albeit Pulling Back A Drop – Despite continued premium takeover activity with Big Pharma/Big Bios, the XBI remains strongly correlated to interest rates. When the recent inflation reports showed worsening figures, Fed Chair Jay Powell kept rates steady with less likelihood of a cut in the very foreseeable future. Hence, rate sensitive stocks like bios are negatively impacted. On the charts, the index continues its move below the 50-day moving average (MA) – keeping profit taking/traders going the other way after a sustained rally from November until the end of February.
Overall biotechs are relatively outperforming driven by the M&A deals, solid fundamentals – several selective strong quarterly updates with impressive launches, faster than expected clinical timelines and also early FDA approvals. Drug stocks, which often behave defensively in this slowing economic environment – have also pulled back with some Medicare/Medicaid pricing issues (and even some renewed whistleblower claims). Last but not least, while bios are always full of news (expected and unexpected), overall we are in somewhat quiet period after a series of medical symposium and investor symposium. Finally, with income taxes due in a geopolitical mess – fund managers are likely on the sidelines after a strong 2025 and start of 2026 with the Iran conflict creating further and lasting uncertainty. Seasonally the group takes a pause this time of year and that appears to be happening – although there are green shoots all over in both MTSL names (e.g., CLDX, PGEN, VKTX, etc.) and bios overall.
The obesity/GLP-1 market – which has led the sector for a few years now – has also taken a major step back, with both the Wegovy pill approvals and upcoming global patent expirations. Recently, we saw recent positive updates for GCPR, ABBV and VKTX – and overall investors are exhausted and confused on the future leaders and the global market potential. That also is enough to signal a less sanguine biotech tape. For our MTSL catalysts — upcoming key clinical data points for WVE (also novel obesity compound) and quarterly launch update for PGEN (RRP treatment) are due this week. We are rather optimistic for both.
CLDX The Ideal M&A Target: Takeover activity, in our view, will continue with those in need of pipeline fills (e.g., ABBV, PFE, AZN, etc.) supported by burgeoning balance sheets. One key thought of ours is that ABBV – which saw an unexpected approval for JNJ’s IL-23 pill last week – is in need another blockbuster – and we believe that CLDX – with its leading KIT-blocking mast cell antibody – and that is now expected to be on the market by 2028 or sooner – would be a perfect fit for the Rinvoq/Skyrizi franchise at a very attractive price. In fact, with most SMID cap acquisitions in the blockbuster new classes coming in at $9-11 billion – with CLDX only at $2 billion market cap – $5 billion to us would still be so cheap. As a reminder, CLDX just announced that the Phase III CSU data will be at least 6 months early – and released in Q4/year end 2026 (instead of H2:2027) – putting that drug and company in play, in our view. Not that anyone believes that nor is there any such rumors on the Street – but with such exceptional de-risked data with a novel MOA in multiple blockbuster indications ahead – we strongly believe that a CLDX buyout is a “when and how much” and not an “if or maybe” anymore.
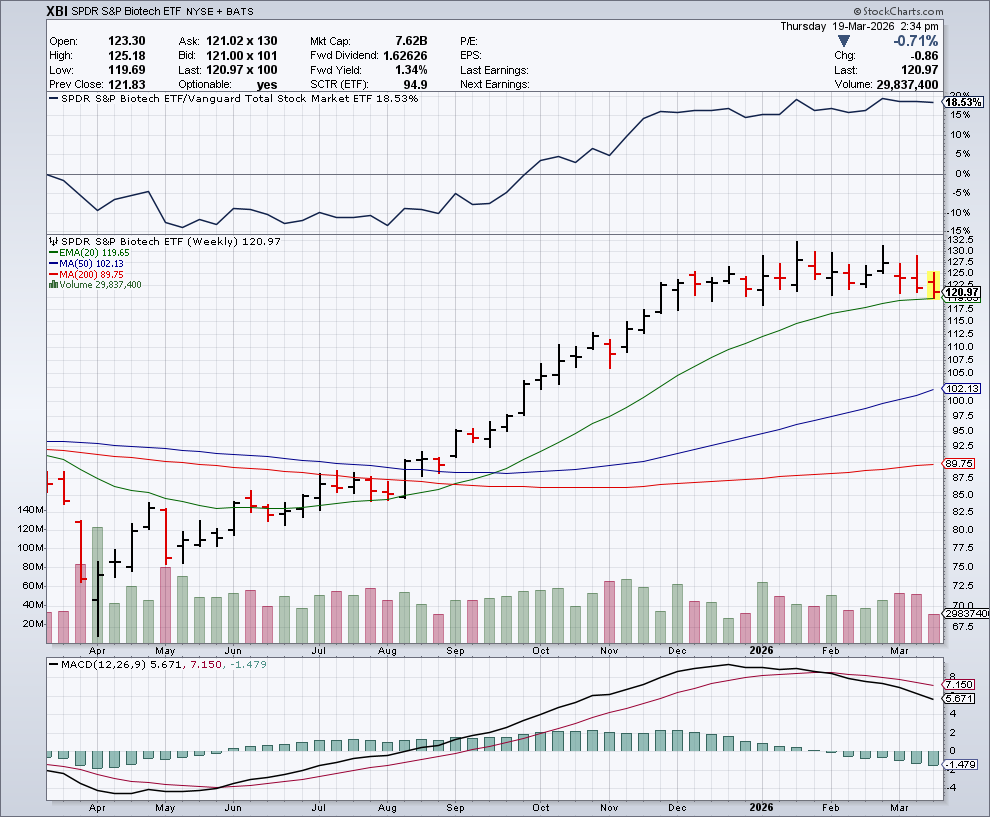
On the charts, the XBI closed at 122 and has an RSI of 44 – a neutral to negative level. The 50-day MA (125) is flattening while the 200-day (106) is still rising. We may remain under pressure for the time being for reasons listed above – less fundamental more macro/technical – but as we’ve seen time and again – things can change rather quickly (e.g., inflation post IRAN, end of the WAR, more M&A, unexpected positive data, etc. could trigger a biotech rally). We continue to recommened our Top MTSL stocks with solid and intact, and in some cases improving, fundamentals during times of quiet market and seasonal pullbacks.
MTSL Events Near Term
- PGEN – 4Q Conf Call March 25 @ 4:30 pm ET
- WVE – 1Q26- WVE-007 Three-month data from the 400mg and six month 240mg follow up data
-
- 1Q26: WVE-006 400mg MAD data from RestorAATion-2 for AATD
- 2Q26: WVE-007-Three month data from 600mg and six month 400mg follow up data
-
- BCYC – 2Q26: AACR Oral presentation for BT5528, April 19, 4:05-4:20 p.m. PT
Clinical Trials Watch
Company Updates
BCYC — BCYC Drops Zele-P, Will Focus on BT5528 & BRC Platform
BCYC provided an update on zelenectide-pevedotin (zele p) and the company has deprioritized the program, as the emerging data from Duravelo-2 is inferior to Padcef. With the shift in strategic focus, BCYC will reallocate its resources to focus on BT5528 and next-generation programs, including its Bicycle Radio-conjugates (BRCs) portfolio. With $628 million in cash, the new runway goes out to 2030 with a 30% reduction in employees. While we are disappointed in the announcement, the move appears to be prudent given the preservation of cash and ability to focus on BT5528 and their Radioconjugate (BRC) platform. With the removal of zele p from the BCYC investment we are lowering our Buy to 10 (from 30) and out Target to 25 (from 60).
BCYC reported a confirmed ORR of 58%/62% at pre-27-week/post-27-week cutoff from dose-selection part of Duravelo-2, which we view as largely comparable to Padcev+pembro’s 67.7% ORR. Notably, only one patient discontinued treatment due to treatment-related AEs (vs. ~35% in EV-302). When combined with Keytruda, the drug stimulated a response at 27 weeks in 58% of people with metastatic disease who have received one or fewer treatment lines. By comparison, Padcev plus Keytruda achieved a response rate of nearly 68%. While zele p may have been safer than Padcef, to date it has not translated into better efficacy.
On the conference call, BCYC said the existing Duravelo-2 trial is no longer considered acceptable as an approval path due to the evolving treatment landscape. Management also indicated that an accelerated approval path may still be possible, but aligning on Ph3 design may take time, and it is not time or resource-efficient to keep the Duravelo-2 running.
Focus Now on BT5528 and BRC portfolio: BT5528 (nuzefatide pevedotin), Ph1 combo data with nivo in mUC will be presented at AACR in April, and BCYC recently started a Phase II trial of single-agent BT5528 in PDAC. On BRC, BCYC will initiate the first company-sponsored BRC program targeting MT-1 using 212-Pb isotope in 2027, with additional imaging data this year.
BCYC recently announced an oral presentation and multiple poster presentations of five abstracts at the American Association for Cancer Research (AACR) Annual Meeting 2026, taking place April 17-22 in San Diego:
- Title: Preclinical assessment of BT5528 anti-tumor activity in cell-line-derived xenograft (CDX) models of head and neck squamous cell carcinoma (HNSCC) Type: Oral presentation
- Session: Advanced Antibody, Conjugate, and Targeted Therapeutic Platforms
- Date and Time: Sunday, April 19, 4:05-4:20 p.m. PT
- Abstract Number: 1325
- Lead Author: Lukas Stanczuk, Ph.D., Bicycle Therapeutics
With BCYC moving away from further development of zele-p with an additional 30% reduction in headcount, BCYC expects to reduce the annual OpEx by 50% to extend the financial runway into 2030 (vs. prior 2028 guidance). We expect BCYC to provide a comprehensive update later this year to showcase its platform/programs with realigned priorities. In our view, the move appears to be prudent given the preservation of cash and ability to focus on BT5528 and their Radioconjugate (BRC) platform. With the removal of zele p from the BCYC investment we are lowering our Buy to 10 (from 30) and out Target to 25 (from 60).
SKYE — SKYE to Test Higher Nimacimab Doses (400mg & 600mg) Data in 4Q2026
Last week on their 4Q conference call, SKYE discussed how they have expanded upon their dose/PK optimization strategy for nimacimab. Importantly, the company is adding a “part C” to the current Phase IIa trial, which is a key de-risking step before Phase IIb begins (likely in 4Q26/1Q27). The start of the Part C expansion study, evaluating 400mg and 600mg IV cohorts is a strategic move designed to answer the central question from the 200mg experience, which is whether the limited monotherapy signal was due to underexposure in peripheral tissues rather than a fundamental limitation of the CB1 pathway. The IV route allows for clean exposure separation to model peripheral engagement for the Phase IIb dose selection while preserving the CNS-sparing safety profile and probably also speeds up the timeline. SKYE plans to report topline out to 16-wks for 400mg and 600mg IV nima to inform go-forward dose. We do not believe the expansion study is powered for efficacy and expect takeaways to focus on safety and PK exposures.
At the lower 200mg dose, the limiting exposure appears to be peripheral tissue (especially adipose) rather than blood. In that framework, the move to higher exposure is meant to test whether the harder-to-reach peripheral compartments can be brought more meaningfully into range, which is the central rationale for Part C. SKYE believes it can push systemic exposure higher while still maintaining a meaningful CNS-sparing margin, which is what makes the higher-dose experiment worth running.
The next step is straightforward and will define the peripheral exposure-response at higher doses through the Expansion Study while using the FDA Type C feedback. The goal would be to then to start the Phase IIb trial with clear dose-selection logic and success criteria across monotherapy and combination development, most likely incorporating the higher 400mg or 600mg dosing. We expect the finalization of the Phase IIb trial design to fully incorporate the FDA feedback utilizing the add-on strategy in GLP-1-experienced adults, ensuring the trial is rigorously powered.
This data could serve as a significant catalyst in 4Q2026 when SKYE plans to report topline out to 16-wks for 400mg and 600mg IV nima to inform go-forward dose. We do not believe the expansion study is powered for efficacy and expect takeaways to focus on safety and PK exposures. We would expect SKYE to move rapidly into a broad Phase IIb program at higher doses as both monotherapy and in combo with GLP-1s.
BioInvest’s John McCamant will be speaking at The MoneyShow’s Master Symposium, April 9-11, at The Diplomat Beach Resort in Hollywood, Florida. Please join him and other financial experts and economists as they come together to discuss new and exciting investing and trading opportunities for 2026. Click the button below for more conference details, the conference schedule and to register for the event.
The Back Page
| Symbol | Company | Orig.Rec. | Current | Target | Recommendation |
|---|---|---|---|---|---|
| ACAD | Acadia | 33.79 | 20.72 | 45 | BUY under $28 |
| ALKS | Alkermes | 10.13 | 28.40 | 55 | BUY under $35 |
| BCYC* | Bicycle* | 43.92 | 4.55 | 25* | BUY under $10* |
| BMRN | BioMarin | 12.68 | 55.03 | 150 | BUY under $100 |
| CLDX | Celldex | 10.50 | 30.03 | 100 | BUY under $75 |
| ESPR | Esperion | 24.42 | 2.60 | 25 | BUY under $10 |
| INCY | Incyte | 5.88 | 92.95 | 108 | BUY under $85 |
| IONS | Ionis | 7.63 | 71.32 | 95 | BUY under $70 |
| MDGL | Madrigal | 17.00 | 454.22 | 625 | BUY under $525 |
| PGEN | Precigen | 34.42 | 3.32 | 12 | BUY under $5 |
| SGMO | Sangamo | 4.77 | 0.34 | 5 | HOLD |
| SKYE | Skye | 17.00 | 0.66 | 12 | BUY under $6 |
| VKTX | Viking | 16.83 | 33.92 | 145 | BUY under $100 |
| VXRT | Vaxart | 8.00 | 0.65 | 8 | BUY under $3 |
| WVE | Wave | 7.58 | 12.12 | 32 | BUY under $22 |
*New recommendation.
THE MODEL PORTFOLIO*
| COMPANY | SHARES OWNED | TOTAL COST | TODAY’S VALUE |
|---|---|---|---|
| Long Positions | |||
| Acadia (ACAD) | 4,750 | 156,557 | 98,420 |
| Alkermes (ALKS) | 3,800 | 88,690 | 107,920 |
| Bicycle (BCYC) | 2,400 | 105,408 | 10,920 |
| Celldex (CLDX) | 14,322 | 120,828 | 430,090 |
| Esperion (ESPR) | 3,316 | 105,316 | 8,622 |
| Incyte (INCY) | 1,229 | 34,817 | 114,236 |
| Ionis (IONS) | 3,087 | 49,123 | 220,165 |
| Madrigal (MDGL) | 2,477 | 16,524 | 1,125,103 |
| Precigen (PGEN) | 9,690 | 76,510 | 32,171 |
| Sangamo (SGMO) | 19,456 | 253,596 | 6,615 |
| Skye (SKYE) | 13,500 | 229,500 | 8,910 |
| Viking (VKTX) | 12,000 | 201,960 | 407,040 |
| Vaxart (VXRT) | 29,687 | 250,000 | 19,297 |
| Wave (WVE) | 30,000 | 227,400 | 363,600 |
| (03/19/26) | Equities: | $2,953,107 | |
| Cash: | $ 109,208 | ||
| PORTFOLIO VALUE: | $3,062,315 |
*The Model Portfolio is designed to reflect specific recommendations. We began the Model Portfolio on 12/23/83 with $100,000. On 4/13/84, we became fully invested. All profits are reinvested. Stocks recommended since then may be equally attractive, but may not be in the Model Portfolio. Transactions and positions are valued at closing prices. No dividends are created, and we don’t use margin. Interest income is credited only on large cash balances.
The Model Portfolio
BENCHMARKS
| NASDAQ | S&P 500 | MODEL | |
|---|---|---|---|
| Last 2 Weeks | -2.9% | -3.3% | 0.4% |
| 2026 YTD | -2.1% | -0.2% | -14.0% |
| Calendar Year 2025 | 20.4% | 16.4% | 50.7% |
| Calendar Year 2024 | 28.6% | 23.3% | 0.1% |
| Calendar Year 2023 | 43.4% | 24.2% | -8.1% |
| Calendar Year 2022 | -33.1% | -19.4% | -12.7% |
| Calendar Year 2021 | 21.3% | 26.9% | -15.2% |
| Calendar Year 2020 | 43.6% | 16.3% | 13.8% |
| Calendar Year 2019 | 35.2% | 28.8% | 10.7% |
| Calendar Year 2018 | 5.7% | 6.6% | 4.5% |
| Calendar Year 2017 | 29.3% | 19.9% | 65.6% |
| Calendar Year 2016 | 7.5% | 9.5% | -29.6% |
| Calendar Year 2015 | -0.1% | -0.1% | 25.1% |
| Calendar Year 2014 | 13.4% | 11.4% | 29.2% |
| Calendar Year 2013 | 38.3% | 29.6% | 103.4% |
BENCHMARKS
New Money Buys
BioInvest
Contact Info
Medical Technology Stock Letter
John McCamant, Editor
Jay Silverman, Editor
Jim McCamant, Editor-at-Large
BioInvest.com
579 Mangels Ave.
San Francisco, CA 94127
Send us an email
Download a PDF of MTSL Issue #1071
©Piedmont Venture Group (2026). Address: 579 Mangels Ave., San Francisco, CA 94127. BioInvest.com. Email: admin@bioinvest.com. Published 24 times a year. Email subscription rates: 1 year – $399; 2 years – $678; 3 years – $898. You may cancel within 48 hours for a full refund. The information and opinions contained herein have been compiled or arrived at from sources believed to be reliable but no representations or warranty, express or implied, is made as to the accuracy or completeness. In no way shall this newsletter be construed as an offer to sell or solicitation of an offer to buy any securities. The publisher and its associates, directors or employees may have positions in, and may from time to time make purchases or sales of, securities mentioned herein. We cannot guarantee and you should not assume that future recommendations will equal the performance of past recommendations or be profitable.



