MTSL Issue 1021
IN THIS ISSUE: SKYE High With CB1 Antibody For Obesity Without the THC/Buzz
Since Last Issue: BTK: 0.79%; NBI: -0.06%; XBI: 5.52%; Model Portfolio: 30.78%
SENTIMENT — “You Ain’t Seen Nothing Yet”
Obesity/MASH Leads BioPharma With More Positive Data From VKTX and Zealand
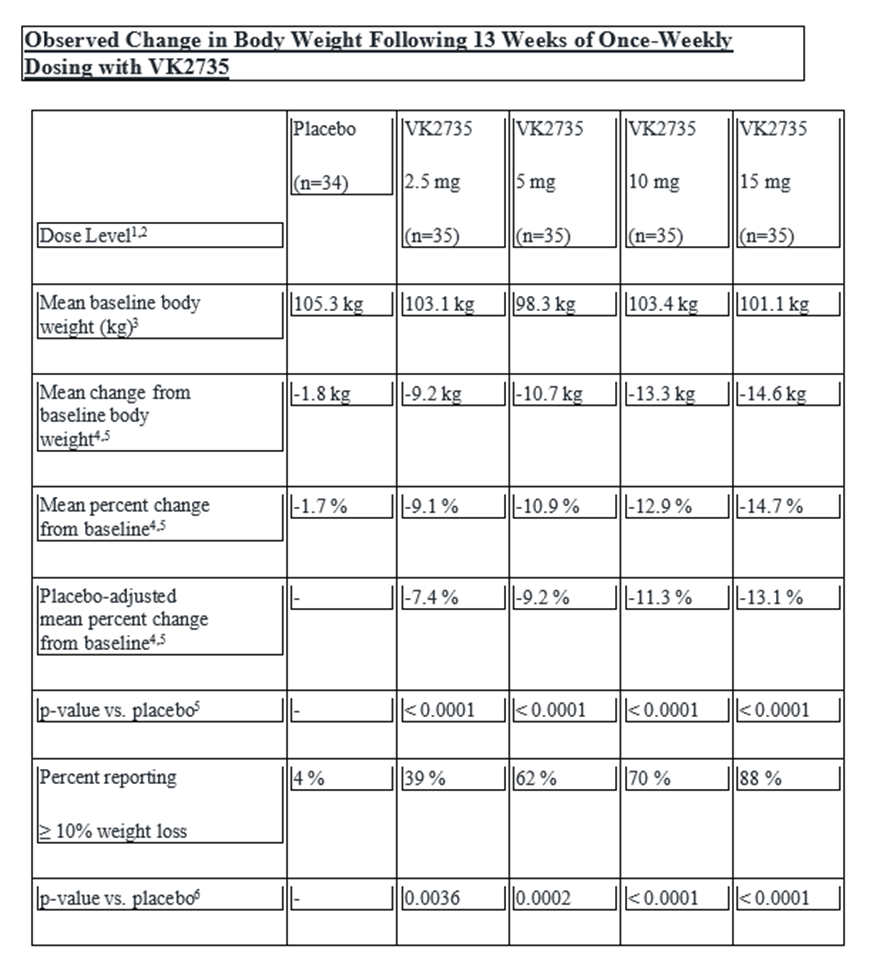
The GLP-1 receptor agonists are transforming the treatment of Type 2 diabetes (T2D) and obesity, and the 11 approved GLP-1’s collectively achieved sales of $36.5 billion in 2023 making it the largest drug class in industry with growth potential in excess of $100 billion. VKTX shares soared this morning when the Company released excellent Phase 2 SubQ data for VK2735 (GLP-1/GIP) showing treated patients lost up to 14.7% of their body weight over 13 weeks, in a clean dose-dependent manner with high statistical significance (p<0.0001). The primary endpoint win is a 13.1 percentage point reduction in weight than the placebo group. In comparison, Eli Lilly’s Zepbound resulted in a 7-8% improvement over a placebo in the Phase 3 study (Surmount-1). Lilly’s retatrutide – which targets all three Gs – led to 12-13% weight loss versus placebo, however with more side effects than the Viking compound, including cardiovascular risk. Investors were led to expect a bar of just 8% placebo-adjusted weight loss for ‘2735 – which has now moved into position as potential Best-in-Class drug.
Zealand Pharmaceuticals announced partner Boehringer Ingelheim’s GLP-1/glucagon survodutide improved metabolic dysfunction-associated steatohepatitis (MASH) versus placebo in a Phase II trial. We remain skeptical regrdaing the third G, glucagon, as it appears to add cardio risk without adding much efficacy.
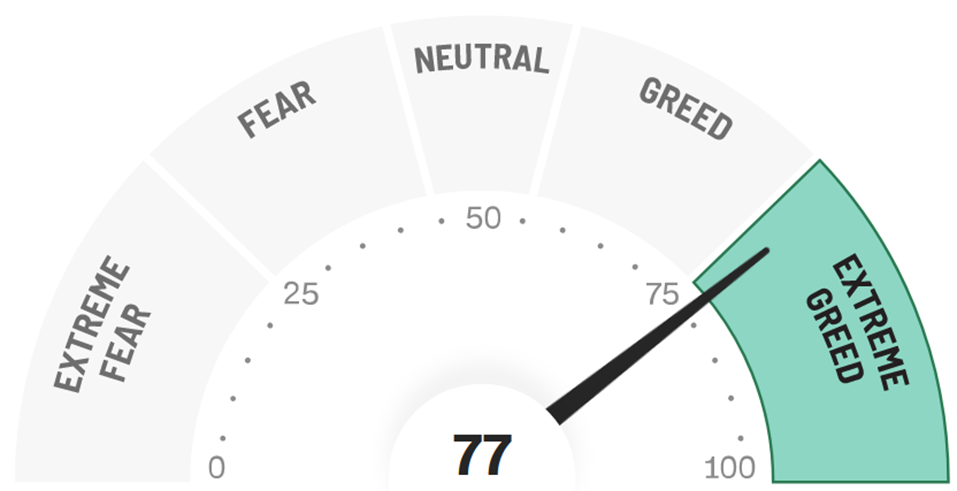
FEAR & GREED – STILL EXTREME
Extreme Greed rules the equity markets as AI enthusiasm ratchets higher following another blowout Q and guidance from Nvidia. For some perspective, the one day market cap gain for NVDA on the announcement of $277 billion is greater than the combined market capitalizations of Pfizer and Bristol Myers! The last week of February started with the S&P Select Biotech Index rallying for a fourth consecutive day to a new high for the year despite no new M&A and a tick up in interest rates. The move represented the 4th best day of the year and seemed technical in nature as the top end of the recent trading range is tested. The S&P 500 has now advanced for 15 of the last 17 weeks, which has only happened one other time in the last 50 years (1989).
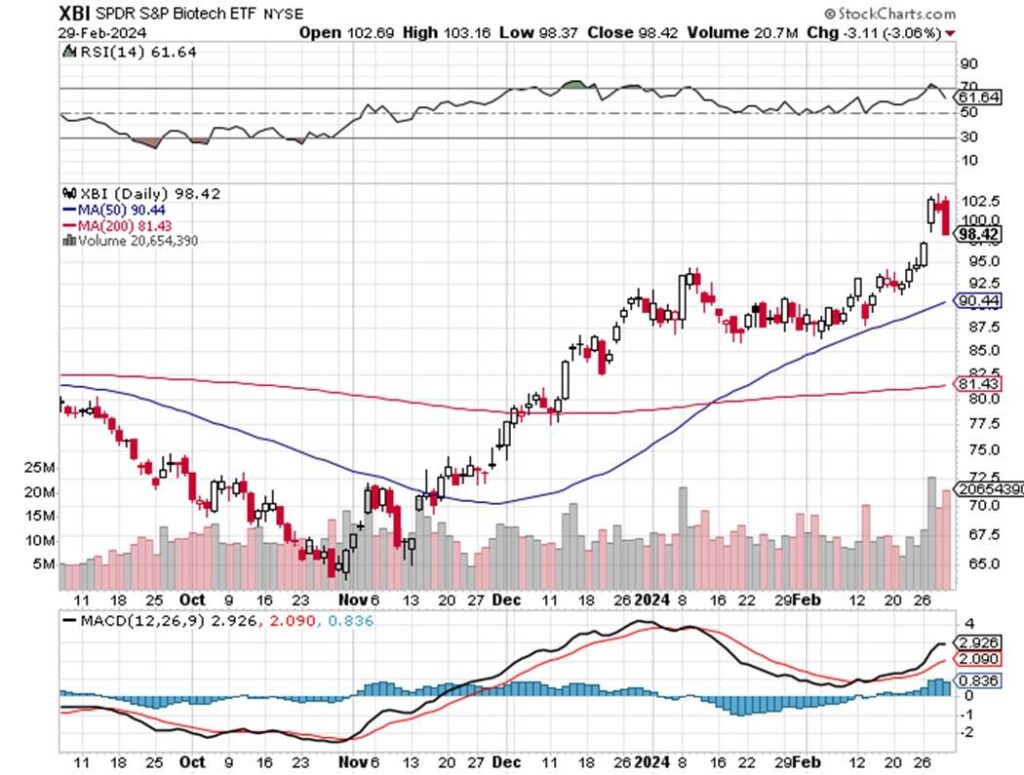
TECHNICALS – XBI HIT OVERBOUGHT LEVELS DRIVEN BY FUNDAMENTALS
The XBI (98) had a whirlwind of volatile performance this week – mostly to the upside – with the index breaking above resistance on the stellar VKTX obesity data, plus CLDX’s CSU update at AAAAI. The biotechs were up on average 15% in February alone. At press time, the sector saw a big drop after the biggest 3-day jump in years coupled with an abundance of follow-on equity offerings. The index briefly touched 106 – it may have been a near term technical sell signal – and that is OK and actually healthy. With premium M&A, excellent clinical results, upcoming FDA approvals and a peaking of interest rates, biotech stocks are back. The volatility is somewhat scary after the huge movement in many stocks, but true biotech investors cannot be unhappy by the recent price action in the sector.
Of course, bankers rush companies to finance (and get the fat fees) and that often leads to a short-term pullback – due to not only dilution but also the fact the deals remove a lot of near term takeover speculations in respective stocks. Nonetheless, cash is often insatiable in this group and the worthy ones with real positive data are able to fund their top compounds and remain independent. With the Big Pharma/Big Drug stocks flush with mountains of cash, they are willing to pay up for novel assets to shore up their long-term pipelines to help offset upcoming patent expirations. Plus, the Street has until now been rewarding both target and acquirers’ stocks. There is no doubt a risk on market occurring (see Bitcoin) and biotechs are also a huge beneficiary of this type of environment. The GLP madness continues – and we expect a ton of more wannabes and copycats.
We are proud of our VKTX recommendation and expect MDGL to gain approval by its PDUFA date (3/14), but the noise will only get louder from here. It is also why we are so aggressive on our CLDX call – it is the ONLY real mast cell inhibitor around with exceptional data and a lot more to come in very large markets. Also, its market cap is relatively small versus CSU alone plus the multiple indications ahead. Currently the XBI 50-day and 200-day moving averages are at 90 and 81, respectively, and the index closed at 98. The RSI, is now just 61-62 down from overbought 72-73 on Tuesday after the VKTX release. We still have some data coming (VKTX oral, MDGL and ESPR PDUFA dates, ACAD data) and the big Cowen Conference this week. So yes its been a good feel mood but after 2-3 years of lagging performance, as the classic rockers Bachman Turner Overdrive sing, “You Ain’t Seen Nothing Yet” – funds are flowing into biotech again.
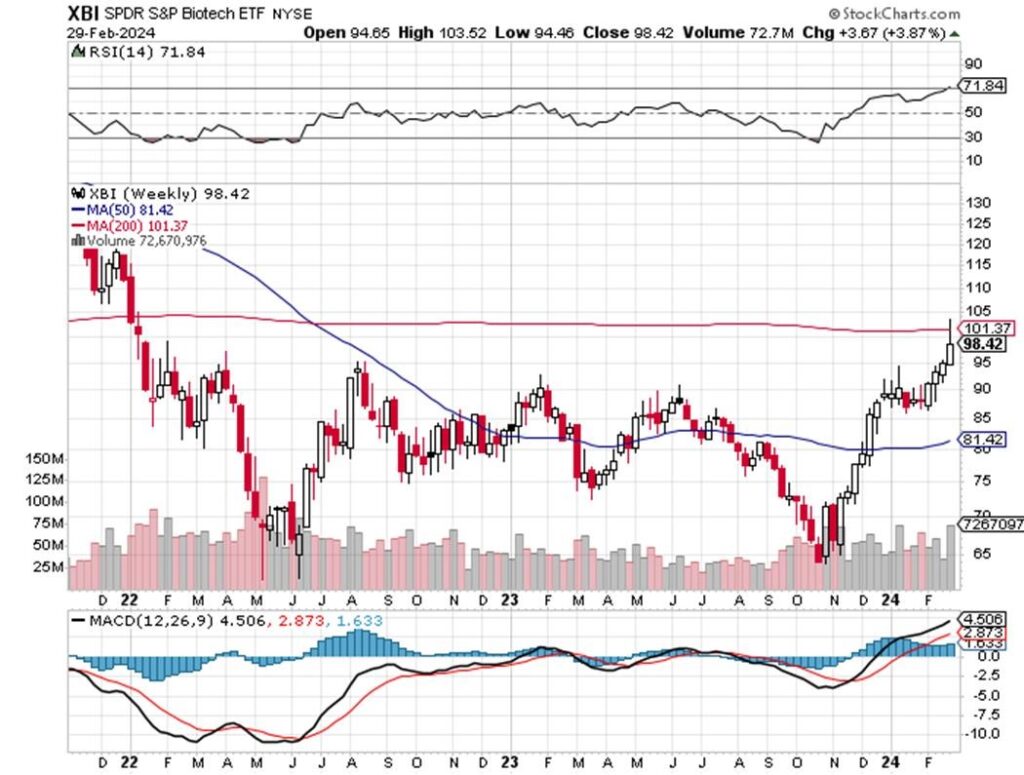
On a weekly basis, the index did in fact blow through the 200-week moving average – the first time since the summer of 2021 – almost 3 years ago. It was a huge move and that was a sell signal in the near-term with the close below at 98. But there is no question the group is strong and in favor despite the sizable one-day of profit taking on Thursday (2/29). It will be interesting to see the fund flows and performance of dedicated funds for February and YTD 2024. There is no doubt that the rise in stock prices coincides with an abundance of large financings (e.g., VKTX is raising >$600 million and CLDX is raising ~$450 million, respectively, with the green shoe) and that depresses shares in the short term. Overall, however, fundamentals and rates are in a sweet spot and the upcoming FDA approval of MDGL’s resmetirom will be widely watched to evaluate the drug and class’ blockbuster potential. It will not be GLP sized, but should nonetheless amass multi-billion dollars in sales. Enjoy the ride. MTSL will keep you abreast and ahead of the catalysts – as Q1 has seen our recommendations lead the biotech sector – notably VKTX and CLDX, with more to come in ALKS, IONS, MDGL – and small cap turnarounds in ESPR and SGMO, too. Lastly, the group remains in M&A mode, so expect certain takeover targets to rally on Fridays with deals often announced on Monday mornings.
MTSL Events Due Near-Term
- VKTX Q1-Phase I Data from Oral Formulation for VK2735 Expected in Q1
- MDGL Q1 – March 14 PDUFA for Resmetirom for NASH
- ACAD Q1 – ADVANCE-2 Phase III study of pimavanserin in Negative Symptoms of Schizophrenia expected Q1
- ESPR Q1 – CLEAR Outcomes Label Expansion PDUFA March 31
- CLDX Q1 – Initiate Phase II study of barzolvilumab in prurigo nodularis (PN)
- PGEN Q2 – Pivotal Phase II RRP data for PRGN-2012 in Q2 which will lead to an FDA filing
Clinical Trials Watch
Company Updates
ACAD — ACAD Down on Lower Daybue Guidance, ADVANCE-2 Up Next
ACAD’s fourth quarter was dominated by Daybue’s sales of $87 million versus the consensus of $83.3 million. Although the company guided a quarter over quarter decrease in Q1 2024 ($76-82 million) based on the seasonal dynamics, we expect sales to continue to grow subsequently. In our view, given the strong interest from both Rett physicians and the patient community there should be a continued growth despite seasonal hiccups in the near term. Additionally, the patient adherence rate was found to be better than what LILAC data showed suggesting the positive benefits of dose titration and efforts to address diarrhea. For Nuplazid, ACAD reported revenue of $143.9 million compared to the consensus of $139.1 million. For the full year 2024, Acadia guided net sales revenue for Daybue between $370-420 million and Nuplazid to $560-$590 million.
The unique mechanism of action (MOA) of pimavanserin has allowed ACAD to focus solely on negative symptoms in the Phase 3 design of ADVANCE-2. Given the landscape of schizophrenia drug development, pimavanserin has a unique position to be marketed solely for the negative symptom patients which might differentiate the drug concerning safety and efficacy.
ADVANCE-2 only includes patients who are stable on positive symptoms, focusing solely on the negative symptoms. Additionally, the 26-week study uses NSA-16, unique to assess negative symptoms. The trial length is also important to make sure that the drug effects are not temporary which is common for antipsychotics used to treat negative symptoms currently. The safety profile is well characterized given that the drug is selective for the serotonin receptors and hence the chances of off-target effects are minimal. In our view, ADVANCE-2 has a decent shot at success given the unique MOA and improved trial design.
BMRN — Rocktavian Sales Almost Non-Existent, Voxzogo Sales Look Great
Rocktavian sales are off to a really slow start and the product was administered to just two patients (one German, one American) in 4Q, generating $2.7 million in net revenue. The numbers so far imply net revenue per patient of $0.8 million and $1.9 million in Germany and the US, respectively. Sales in Italy may soon commence following recent reimbursement approval. Analysts estimates have also come down substantially and predict BMRN will be lucky to generate even $40 million in revenue in 2024 for the Hem A gene therapy. Maybe doctors are waiting for the SGMO/Pfizer product which should be approved by year end and in our view is a superior gene therapy.
On a more positive note, Voxzogo’s strong trajectory continued through 4Q following October’s FDA label expansion to younger patients.
Revenue was $146M (+18% Q/Q), with better-than-anticipated supply providing a boost. Of new starts in 4Q, ~70% were <5 years old. At year-end, 2,613 children (+127% vs. YE22) were on treatment across 41 markets. We anticipate a Hypochondroplasia pivotal trial to begin mid-2024 following current run-in, and pivotals in other short stature conditions to begin this year pending discussions with regulators.
CLDX — (2/25/24 Update) Barzo Shines At AAAAI Meeting
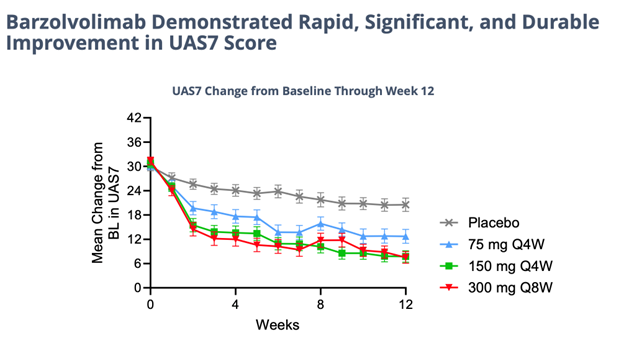
On further inspection of the Phase II 12-week CSU data, barzolvolimab’s wide therapeutic window continues to support its Best-In-Class compound profile. The urticaria market is extremely underestimated and we expect barzo forecasts will rise significantly from here. BUY
Unprecedented Efficacy…
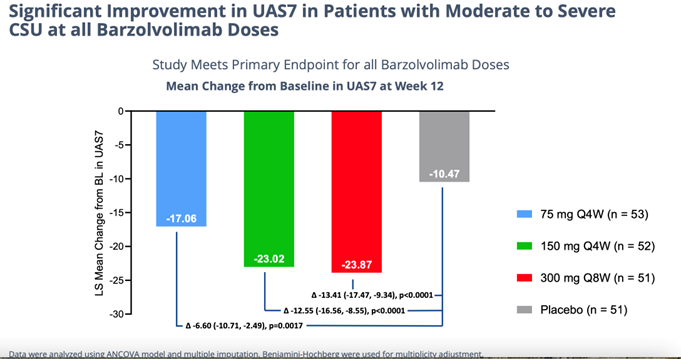
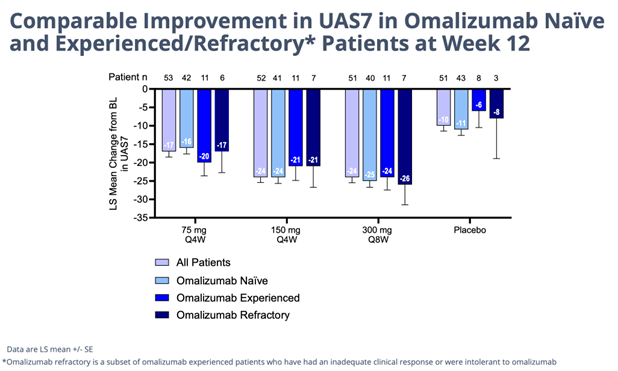
Details of the successful, 208-patient 12-week study study of barzo in CSU were presented in an oral Late-Breaker trial on Saturday at the AAAAI meeting. The drug’s novel advantageous mechanism of action is evident by the consistent, superior human clinical data delivered last November and shown again this weekend. In a highly severe patient group barzo showed a rapid onset of action (~2 weeks); durable, total disease control of CSU (but now in all forms of urticaria) – UAS7=0 was observed in 51% of patients in the 150mg Q4W and in 37.5% of patients in the 300 mg Q8W groups compared to 6.4% in the placebo group); and a more than acceptable safety profile. Moreover, the CLDX drug works just as good in Xolair (omalizumab) refractory, experienced and naive patients. Moreover, as in previous studies the responses with barzo deepen over time. Even though the data below is exceptional at just 12 weeks in and of itself, though not necessary to maintain its Best-in-Class profile, by 52 weeks the efficacy could possibly get even better.
…Supported By A Clean Safety Profile
One of the major investor (and bears/shorts) concerns coming into the meeting was the potential for neutropenia to be a limiting factor in barzo’s A/E profile. We have seen the efficacy presented above last November, but other than CLDX saying the safety was clean, until this weekend we had not yet seen the details. Of the 153 patients on Barzo in this study, just 2 patients discontinued due to neutropenia (Grade 3), with only 1 of those being confirmed by lab tests. However, Dr. Mauer quickly and effectively pointed out that one patient had pre-existing propensity for thrombocytopenia (not neutropenia) and the both of the patients briefly dropped below the pre-specified 1000 neutrophil count and had begun their recovery back above the threshold (one did it before and the other after the cutoff date). In other words, less than 1% of treated patients showed any sign of severe neutropenia and that resolves entirely over time and with drug discontinuation. According to both Drs. Maurer and Kaplan – barzo is a safe drug – and a much safer alternative than is currently being used to treat urticaria (e.g., steroids, cyclosporine, Xolair, the BTK inhibitors under development). In addition, in previous studies with the REGN/SNY blockbuster Dupixent, the rate of neutropenia in atopic dermatitis studies was over 4%. Both KOLs at the meeting easily dismissed neutropenia as a problem with barzo and reminded investors that every patient is pre-tested for these counts before beginning treatment anyway. In addition, Genentech’s Xolair – the only approved drug in the U.S for urticaria – carries a black box warning for anaphylaxis. Although there is always some risk in larger and longer term studies, at this stage of development the Barzo side effect profile looks very clean, easily refuting the bear/short call going into the meeting.
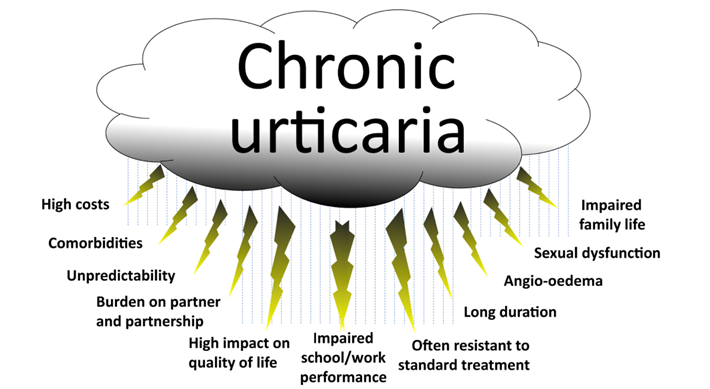
The Urticaria Market Is Much Bigger Than People Think
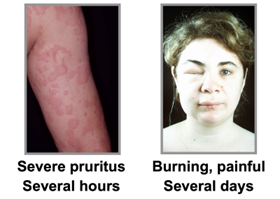
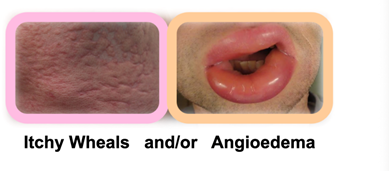
Both Mauer and Kaplan showed proof that urticaria and that chronic urticaria is a more devastating disease than most people are aware of, and with new treatments like Barzo this is about to change. Patients with CSU suffer constantly (with itch wheels, etc.), have an awful quality of life, and there is also a silence about the condition due to the physical stigma and lack of adequate treatments. Below we show some of examples the physical symptoms of urticaria. The vast number of CSU studies by Big Pharma and biotech firms suggest a huge market is approaching. Just wait until the advertising begins when new treatments like Barzo are approved.
While some older U.S. numbers have suggested a prevalence of 0.1-0.2% of the population, Mauer pointed out a more recent and vigorous epidemiology study of CSU done in China last year suggest a 2.6% prevalence and that the disease is even more severe in Asian and Latin American populations than in the US/EU.
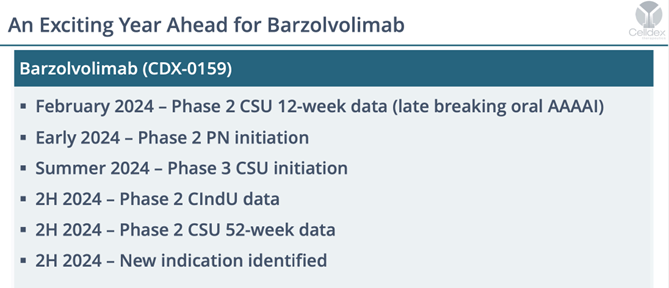
What’s Next for Barzolvilumab? A Whole Lot
The remainder of 2024 will include some major clinical catalysts including the start of Phase III studies that will support a BLA filing (summertime). Also, very soon we will get the start of Phase II prurigo nodular (PN) trials – another underestimated illness that Barzo has shown incredible (albeit small study) efficacy. In the second half, the Phase II CindU (cold induced urticaria) will come due and the all important 52-week data from this CSU trial will be out in H2 as well (likely by the November meetings).
Last But Not Least Is The 50->100 Conditions That Mast Cells Play A Key Role In
For a while now, we have pounded the table that Barzo will be a blockbuster drug. The AAAAI meeting further supports our hyphothesis. CLDX will announce a fifth clinical indication sometime in the second half of this year. but there are so many more. Dr. Kaplan, for example, yesterday mentioned that mast cells are over activated in synovial fluids of rheumatoid arthritis patients. Last week, we put out a note about food allergies. See our MTSL initiation report from August 2020. This is just the beginning.
ESPR — ESPR Reports Solid Q4 Revenue, March 31st PDUFA Up Next
ESPR recently reported revenue of $32.3 million for the three months ended December 31, 2023, and $116.3 million for the full year ended December 31, 2023, compared to $18.8 million and $75.5 million for the comparable periods in 2022. U.S. net product revenue was $20.8 million for the three months ended December 31, 2023, and $78.3 million for the full year ended December 31, 2023, compared to $15.0 million and $55.9 million for the comparable periods in 2022, driven by retail prescription growth of 44% and 30%. Collaboration revenue was $11.5 million for the three months ended December 31, 2023, and $38.0 million for the full year ended December 31, 2023, compared to $3.9 million and $19.6 million for the comparable periods in 2022, driven by increased royalty revenue and tablet shipments to international partners.
ESPR’s pending applications with the FDA and EMA are progressing extremely well and are on track with an FDA PDUFA date of March 31, with an expected decision by the EMA on the European label likely coming in the second quarter. The company is also making progress with the payers, with some payers already making changes in anticipation of the new label. ICER just recently came out and made reference to NEXLIZET as being a cost-effective therapy and that it should essentially be made available on all formularies, which will definitely help payers reimburse.
SKYE — SKYE High With CB1 Antibody For Obesity Without the THC/Buzz
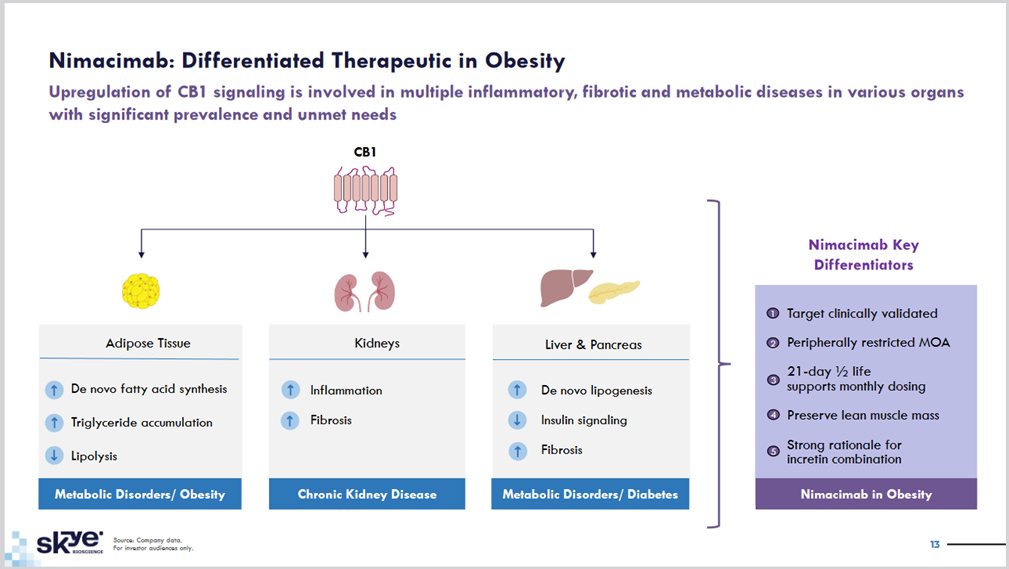
We are recommending Skye Bioscience (SKYE) as our next investment opportunity in metabolic disease; obesity and NASH/MASH. Skye is developing a peripherally-restricted CB1 antibody — nimacimab — to treat obesity by restoring leptin sensitivity and increasing fat metabolism while avoiding the CB1 induced THC/buzz. We believe this mechanism of action (MOA) is validated by Novo Nordisk’s recent acquisition of privately-held Inversago for $1.075 billion. SKYE plans to shortly initiate a Phase II study of nimacimab +/- semaglutide in obese patients in mid-2024 with preliminary data due in 1Q2025. We believe the company’s unique approach of developing a selective large molecule antibody with muscle-sparing weight loss and no THC/buzz side effects has significant potential. The key is that large molecules like antibodies are denied entry into the brain by the blood brain barrier (BBB), while CB1/THC small molecules easily access the brain and lead to significant impairment. In our view, the nimacimab Phase II obesity trial has potential to serve as proof of concept and positive data would be a huge catalyst for the stock. We are recommending SKYE as a BUY under 24 with a TARGET PRICE of 40.
Why CB1 Has Potential in Metabolic Disease
SKYE’s nimacimab has a unique mechanism of action that selectively targets the endocannabinoid system (ECS) to treat obesity/metabolic disorders and glaucoma. The primary role of the ECS is to introduce, accumulate, and store energy in the body enabled by activity of cannabinoid type-1 receptor (CB1) in the brain and peripheral tissues. Recent work has uncovered that CB1 inhibition in the periphery can regulate leptin sensitivity, likely due to signaling effects on adipocytes. CB1 activation stimulates adipogenesis and lipogenesis, leading to impaired mitochondrial function, whereas genetic and pharmacological inhibition of CB1 increases mitochondrial biogenesis. Consequently, body weight loss induced by administration of CB1 antagonists is largely due to increased energy expenditure and activation of lipolysis and fatty acid oxidation. Lipolysis occurs in brown adipose tissue, which operates as a source of energy, as opposed to white adipose tissue, which serves as a stable longterm store of fat. It is currently hypothesized that the tone of CB1 activity determines the balance of white and brown fat, and as a result, metabolic rate. The upcoming Phase II trial will assess body composition and may demonstrate that nimacimab spares muscle loss due to its fat “browning” mechanism. In our view, Skye’s approach of developing a CB1 antibody with muscle-sparing weight loss and no psychiatric side effects has block buster potential.
Novo Nordisk’s Wegovy (semaglutide) and Eli Lilly’s Zepbound (tirzepatide) are revolutionizing weight management in obese patients with GLP-1s which were originally approved for Type 2 diabetes. Now these drugs are even showing cardiovascular benefit due to weight loss and should eventually show a long term mortality benefit from the improved CV profile. While Zepbound was only recently launched, Wegovy revenues were $4.7 billion in 2023, and the class is projected to grow to $20 billion by 2030! Despite unprecedented efficacy and commercial success, there remains room for improvement over currently weekly injectable GLP-1 and GIP agonists for the treatment of obesity. In most cases, GLP-1 monotherapy does not get patients to a healthy BMI, and there is the potential for even greater weight loss through combination therapy. Preclinical evidence suggests that targeting both GLP-1 and CB1 mechanisms has an additive effect on weight loss, but also provides a restoration of insulin sensitivity that is not seen with GLP-1s alone. GLP-1s are not the end all for obesity as real world observations estimate a 50% dropout. Lastly, CB1 make make GLP-1s more tolerable as CB1/THC is known to have a beneficial effect on nausea.
SKYE is planning a Phase II study of nimacimab in 200 non-diabetic obese patients (BMI≥30) or overweight patients (BMI≥27) with at least one weight-related co-morbidity. 50 patients each will be randomized to 200mg weekly subQ nimacimab, 2.4mg weekly subQ semaglutide, the combination, or placebo for 26 weeks with 12-week follow-up. (See Slide Below.) The primary endpoint is percent change in weight at week 26. Secondary endpoints include changes in waist circumference, body composition, fasting triglycerides, cholesterol and A1c. This design will enable Skye to determine the weight loss effect of monotherapy nimacimab vs. semaglutide, as well as potentially additive effects of combination with semaglutide. Beyond weight loss, we are particularly focused on differences in fat/muscle body composition between the arms. We anticipate the nimacimab arms may show improved muscle preservation due to its mechanism of fat browning. In preclinical studies, CB1 inhibition showed significant weight loss without loss of muscle mass over a 28-day period. SKYE anticipates finalizing design and initiating the Phase II trial shortly.
SKYE recently raised $50 million in a PIPE financing which was co-led by a life sciences-focused investor and 5AM Ventures, with participation from Ally Bridge Group, Sphera Healthcare, Altium Capital, Driehaus Capital Management and other institutional investors. We believe that The Baker Brothers are also new investors and will appear in future filings. In our view, this is smart money and further de-risks the SKYE investment. The company is currently thinly traded on the pink sheets and will be uplisted to NASDAQ within a few weeks.
Skye is also conducting a Phase IIa study of eye drop formulation of CB1 agonist SBI-100 for the treatment of glaucoma with data expected in 1H:24. While positive data would help the investment story, our primary reason for recommending SKYE is the intriguing potential of a peripherally-restricted CB1 antibody, nimacimab, to treat obesity by restoring leptin sensitivity and increasing fat metabolism while avoiding neurological impairment/buzz.
Up Next For SKYE
- Report Phase IIa data on SBI-100 in glaucoma in H1:24
- Initiate Phase II obesity study of nimacimab in mid-24
- Initiate Phase IIb study of SBI-100 in glaucoma in H2:24
- Report interim Phase II obesity data on nimacimab by Q1:25
- Report final Phase II obesity data on nimacimab by Q4:25
The upcoming Phase II study is designed to demonstrate proof-of-concept for nimacimab’s unique MOA and to showcase a potentially differentiated safety and efficacy safety profile. Positive Phase II data could result in a nimacimab partnership or even a SKYE acquisition by BigPharma. We believe the company’s unique approach of developing a selective large molecule antibody with muscle-sparing weight loss and no THC/buzz side effects has significant potential. The key is that large molecules like antibodies are denied entry into the brain by the blood brain barrier (BBB), while CB1/THC small molecules easily access the brain and lead to significant impairment. In our view, the nimacimab Phase II obesity trial has potential to serve as proof of concept and positive data would be a huge catalyst for the stock. We are recommending SKYE as a BUY under 24 with a TARGET PRICE of 40.
VKTX — (2/27/24 Update) VK2735 Delivers The Best 3-Month Obesity Data Yet With 14.7% Weight Loss; Highly Successful Proof of Concept Result Could be Best-In-Class Compound In The Biggest Drug Market In History – Raising BUY to 100, TARGET to 145
VKTX shares soared this morning when the Company released excellent Phase 2 SubQ data for VK2735 showing treated patients lost up to 14.7% of their body weight over 13 weeks, in a clean dose-dependent manner with high statistical significance (p<0.0001). The primary endpoint win is a 13.1 percentage point reduction in weight than the placebo group. In comparison, Eli Lilly’s Zepbound resulted in a 7-8% improvement over a placebo in the Phase 3 study (Surmount-1). Lilly’s retatrutide – which targets all three Gs – led to 12-13% more weight loss versus placebo, however with more side effects than the Viking compound, including cardiovascular risk. Investors were led to expect a bar of just 8% placebo-adjusted weight loss for ‘2735 – which has now moved into position as a potential Best-in-Class drug. One apparent advantage of the Viking compound is the potency and durability – it may be administered once-a-month versus weekly or every other week for the current market leaders. In addition, another key catalyst to watch for by the end of Q1 is the oral version of ‘2735 with an expectation for a 1-3% placebo-adjusted weight loss. In our view, this Phase II SubQ data de-risks the upcoming Phase I data for ‘2735. Oral administration (i.e., a pill) is a huge advantage for the class – in particular as a long-term maintenance option – and we are raising our BUY to 100 (from 28) and our TARGET to 145 (from 45).
The Impressive Data:
Safety Looks Great
Regarding safety and tolerability, the data for ‘2735 continues to look generally clean with expected AEs (i.e., mostly GI disturbance) similar for the GLP class as a whole. Discontinuations appear balanced between the treatment and placebo groups, with 13% of patients on ‘2735 discontinuing the study compared to 14% of those on placebo. There appeared to be a slight dose effect in the ‘2735 group. 92% of drug related TEAEs were characterized as mild-moderate to severity, and the majority (95%) were GI-related including elevated nausea (43% vs 20% on pbo) and vomiting (18% vs. 0% on pbo). This common side effect was generally observed early in the course of treatment, in line with other incretins.
The Biggest Drug Class Ever and Continues To Jump Exponentially
The GLP-1 receptor agonists are transforming the treatment of Type 2 diabetes (T2D) and obesity, and the 11 approved GLP-1Ra’s collectively achieved sales of ~$36.5 billion in 2023. With only one oral peptide approved, Rybelsus (semaglutide), the rest of the approved drugs are administered via subcutaneous injection.vObesity could be the largest therapeutic category of all time and approved GLP-1 sales have driven Eli Lilly ($727 billion market cap) and Novo Nordisk ($545 million market cap) to become the two largest pharma companies by market cap, with some suggesting LLY will be the first $-Trillion pharma company. These market leaders are solidifying their positions via M&A with Lilly buying Versanis (novel early-stage anti-obesity mechanism) in 2023 for $1.9 billion and Novo buying Catalent for $11 billion to boost their injectable GLP-1 peptide manufacturing capacity. As a result of the absolute size of the market, additional Big Pharma companies desire to enter the obesity space, and just last year Roche acquired Carmot Therapeutics (3 obesity assets) for $3.1 billion and AstraZeneca acquired an oral small molecule GLP-1Ra from Eccogene for $2.1 billion. In our view, the new ‘2735 data means that VKTX could be the next even after today’s significant move as Big Pharma is chasing scarce assets.
Next Steps – Pipeline Optionality & M&A Potential
VKTX is planning to meet with the FDA mid-year and is currently designing another Phase IIb trial as the next clinical step. Given the promising results, we believe VKTX has multiple options to explore additional dosing regimens and further optimize the safety/efficacy profile and improve patient experience. The next catalyst also due this quarter is the oral ‘2735 data with an expectation for a 1-3% placebo-adjusted weight loss. In our view, this Phase II SubQ data de-risks the upcoming Phase I data for ‘2735. A pill version will ge a huge advantage for the class (many know that Pfizer dropped its lead oral GLP compound late last year).
We are raising our BUY to 100 (from 28) and our TARGET to 145 (from 45) as a reflection of today’s broadly successful ‘proof of concept data for ‘2735 that has what we believe has Best-in-Class potential. We also expect Phase IIb data for VK2809 NASH (VOYAGE) histology data in H1:24 and the upcoming resmetirom FDA approval (PDUFA 3/14, from MTSL Recommendation MDGL) as additional near-term catalysts for VKTX. The GLPs have rapidly become the biggest drugs in history – they have added roughly $500 billion – $1 trillion in market cap to the leaders in just ~2 years – and therefore the post-data VKTX market cap of $8.5 billion market does not look expensive at all. (After the close, VKTX is raising what appears to be an easily oversubscribed ~$350-400 million secondary offering.)
The Back Page
| Symbol | Company | Orig.Rec. | Current | Target | Recommendation |
|---|---|---|---|---|---|
| ACAD | Acadia | 33.79 | 23.24 | 45 | BUY under $28 |
| ALKS | Alkermes | 10.13 | 30.20 | 55 | BUY under $35 |
| BCYC | Bicycle | 43.92 | 25.09 | 75 | BUY under $50 |
| BMRN | BioMarin | 12.68 | 86.28 | 150 | BUY under $100 |
| CLDX | Celldex | 10.50 | 48.06 | 100 | BUY under $75 |
| ESPR | Esperion | 24.42 | 2.51 | 25 | BUY under $10 |
| INCY | Incyte | 5.88 | 58.36 | 108 | BUY under $85 |
| IONS | Ionis | 7.63 | 45.21 | 65 | BUY under $50 |
| MDGL | Madrigal | 17.00 | 241.70 | 400 | BUY under $300 |
| PCRX | Pacira | 15.78 | 29.72 | 100 | BUY under $80 |
| PGEN | Precigen | 34.42 | 1.54 | 12 | BUY under $5 |
| SGMO | Sangamo | 4.77 | 1.16 | 5 | BUY under $2 |
| TCRT | Alaunos | 8.00 | 2.09 | 5 | HOLD |
| VKTX* | Viking* | 16.83 | 77.05 | 145* | BUY under $100* |
| VXRT | Vaxart | 8.00 | 1.22 | 8 | BUY under $3 |
*New recommendation.
THE MODEL PORTFOLIO*
| COMPANY | SHARES OWNED | TOTAL COST | TODAY’S VALUE |
|---|---|---|---|
| Long Positions | |||
| Acadia (ACAD) | 4,750 | 156,557 | 110,390 |
| Alkermes (ALKS) | 3,800 | 88,690 | 114,760 |
| Bicycle (BCYC) | 2,400 | 105,408 | 60,216 |
| Celldex (CLDX) | 15,832 | 174,993 | 688,315 |
| Esperion (ESPR) | 3,316 | 105,316 | 8,323 |
| Incyte (INCY) | 1,229 | 34,817 | 71,724 |
| Ionis (IONS) | 3,087 | 49,123 | 139,563 |
| Madrigal (MDGL) | 3,127 | 69,980 | 695,371 |
| Pacira (PCRX) | 2,375 | 63,887 | 70,585 |
| Precigen (PGEN) | 9,690 | 76,510 | 14,923 |
| Sangamo (SGMO) | 19,456 | 253,596 | 22,569 |
| Alaunos (TCRT) | 26,125 | 166,100 | 54,601 |
| Viking (VKTX) | 12,000 | 201,960 | 924,600 |
| Vaxart (VXRT) | 29,687 | 250,000 | 36,218 |
| (02/29/24) | Equities: | $3,012,159 | |
| Cash: | $ 237,884 |
||
| PORTFOLIO VALUE: | $3,250,043 |
*The Model Portfolio is designed to reflect specific recommendations. We began the Model Portfolio on 12/23/83 with $100,000. On 4/13/84, we became fully invested. All profits are reinvested. Stocks recommended since then may be equally attractive, but may not be in the Model Portfolio. Transactions and positions are valued at closing prices. No dividends are created, and we don’t use margin. Interest income is credited only on large cash balances.
*Model Portfolio Update: Will purchase $235,000 of SKYE.
The Model Portfolio
BENCHMARKS
| NASDAQ | S&P 500 | MODEL | |
|---|---|---|---|
| Last 2 Weeks | 1.2% | 1.3% | 30.8% |
| 2024 YTD | 6.0% | 5.4% | 37.8% |
| Calendar Year 2023 | 43.4% | 24.2% | -3.5% |
| Calendar Year 2022 | -33.1% | -19.4% | -12.7% |
| Calendar Year 2021 | 21.3% | 26.9% | -15.2% |
| Calendar Year 2020 | 43.6% | 16.3% | 13.8% |
| Calendar Year 2019 | 35.2% | 28.8% | 10.7% |
| Calendar Year 2018 | 5.7% | 6.6% | 4.5% |
| Calendar Year 2017 | 29.3% | 19.9% | 65.6% |
| Calendar Year 2016 | 7.5% | 9.5% | -29.6% |
| Calendar Year 2015 | -0.1% | -0.1% | 25.1% |
| Calendar Year 2014 | 13.4% | 11.4% | 29.2% |
| Calendar Year 2013 | 38.3% | 29.6% | 103.4% |
BENCHMARKS
New Money Buys
BioInvest
Contact Info
Medical Technology Stock Letter
John McCamant, Editor
Jay Silverman, Editor
Jim McCamant, Editor-at-Large
BioInvest.com
579 Mangels Ave.
San Francisco, CA 94127
Send us an email
Download a PDF of MTSL Issue #1021
©Piedmont Venture Group (2024). Address: 579 Mangels Ave., San Francisco, CA 94127. BioInvest.com. Email: admin@bioinvest.com. Published 24 times a year. Email subscription rates: 1 year – $399; 2 years – $678; 3 years – $898. You may cancel within 48 hours for a full refund. The information and opinions contained herein have been compiled or arrived at from sources believed to be reliable but no representations or warranty, express or implied, is made as to the accuracy or completeness. In no way shall this newsletter be construed as an offer to sell or solicitation of an offer to buy any securities. The publisher and its associates, directors or employees may have positions in, and may from time to time make purchases or sales of, securities mentioned herein. We cannot guarantee and you should not assume that future recommendations will equal the performance of past recommendations or be profitable.