January 31, 2022
BIOINVEST NEWS: Madrigal (MDGL)
Breaking News: Resmetiron Delivers Positive Phase III MAESTRO-NAFLD-1 Data
One of the few broadly positive binary events in biotechnology of late, Madrigal’s best-in-class NASH compound is well on its way with de-risked, late-stage clinical results. With this wholly-owned compound heading to blockbuster status, MDGL remains a Top Pick for 2022.
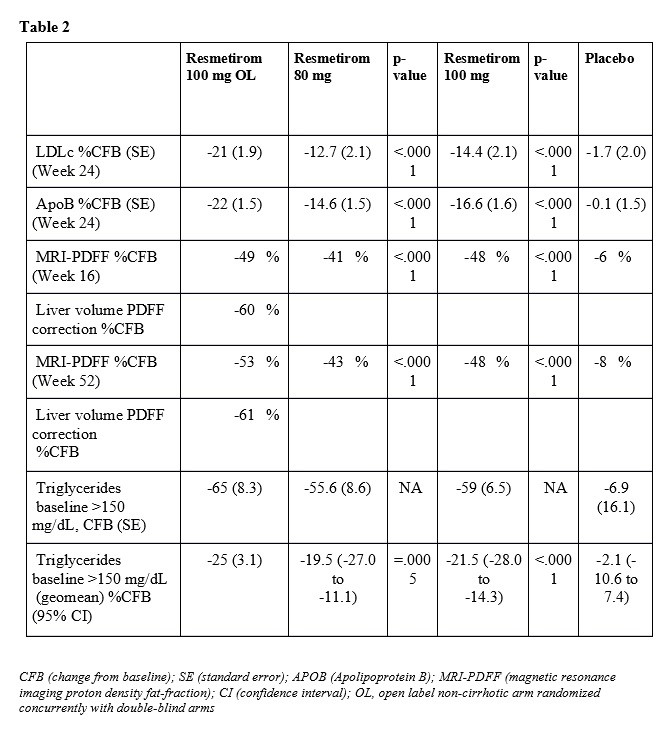
In sum, all primary and key secondary endpoints from the double-blind placebo-controlled 969-patient MAESTRO-NAFLD-1 safety study were achieved and demonstrate that resmetirom:
- Was safe and well-tolerated at 80 and 100 mg in patients treated for 52 weeks
- Provided highly significant (p-value .0001) and clinically relevant reductions in liver fat as measured by magnetic resonance imaging proton density fat-fraction (MRI-PDFF)
- Significantly reduced atherogenic lipids, including LDLc, apolipoprotein B and triglycerides
The increased enrollment of patients into the safety database is consistent with meeting regulatory guidance for chronic disease therapies.
MDGL will continue to generate data from the MAESTRO-NAFLD-1 study, with future readouts of safety, additional biomarkers and non-invasive measures of liver fibrosis, over the next several weeks by our forecast. The Company intends to provide at least one additional public disclosure prior to publication/presentation at a major medical meeting — EASL is in June and is the next major liver disease conference. In addition, the Company is still analyzing liver enzyme data. Despite the widely positive data, investors in this terrible biotech tape want the fibrosis and liver enzymes results – hence the muted stock price response. These results were slightly impacted by COVID. However, just as the Phase II data supported the Phase III efficacy and safety delivered above and the multiple statistically significant endpoints just released, we expect both the fibrosis and liver enzyme data are highly likely to be positive.
While we await further analyses and disclosures from the MAESTRO-NAFLD-1 trial, today’s data are quite impressive and in our view, serve to further de-risk the upcoming 52-week biopsy data from the Phase III MAESTRO-NASH trial expected in Q3:22. By positioning resmetirom for success in both NASH/NAFLD which have no current treatments, MDGL is pioneering the future of medicine by using non-invasive MRI to diagnose disease and then treat with a pill (resmetirom) that targets the source of NASH/NALFD and preventing it from spiraling into death from either a cardio event or liver disease.
Market expectations for NASH drugs has all but disappeared with the failure of multiple compounds over the past several years, and have gotten even lower with the current biotech tape. But that has not been the case with Madrigal, especially after today’s news. (If one recalls, MDGL stock exceeded $300 per share when its positive Phase I/II data were released). With the de-risked data and still wholly-owned, the takeout potential of Madrigal by a Big Pharma/Big Biotech player also rises substantially. BUY