June 26, 2022
BIOINVEST NEWS: Madrigal (MDGL)
Breaking News: MDGL’S Resmetirom Stars at EASL
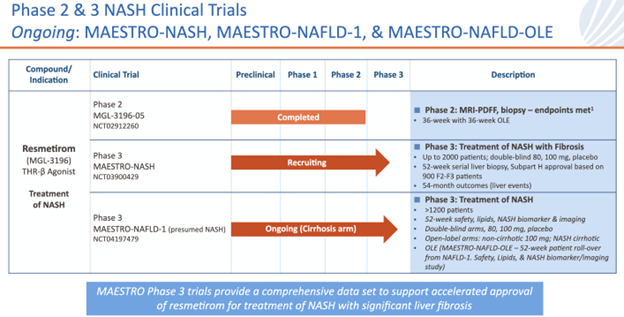
MDGL held a two hour conference call after all presentations at EASL to discuss the outstanding resmetirom data presented at yesterday. The Company focused on the 969-patient MAESTRO-NAFLD-1 safety study which showed the compound to have an excellent safety profile along with excellent efficacy. In addition, the Company presented a 105-patient open-label trial in well-compensated cirrhosis patients that showed an excellent safety profile and both reduced liver and spleen volume. The two trials above combined with the soon to read out MAESTRO-NASH-1 Phase III trial mean that MDGL will have tested resmetirom in chronic liver disease patients at three different stages of disease – NAFLD patients are the least sick, followed by NASH patients, and compensated cirrhosis patients being the most advanced. This is far and away the most comprehensive drug development program in the chronic liver disease space and bodes very well for the first FDA approval and then broad market penetration by resmetirom.
Importantly, MDGL continues to use every non-invasive means possible to measure liver disease efficacy, including routine blood tests and various types of MRIs. This is the “real world” treatment of chronic liver disease which rarely requires a liver biopsy. Current FDA guidelines require liver biopsies for NASH drug approval and MDGL has complied despite their modest use in “real world” diagnosis and treatment of NASH/chronic liver disease. MDGL will file resmetirom for FDA approval via a Subpart H NDA(Subpart H – Accelerated Approval of New Drugs for Serious or Life-Threatening Illnesses; https://www.ecfr.gov/current/title-21/part-314/subpart-H) in early 2023 with close to 2,000 treated patients from two Phase III trials; MAESTRO-NAFLD-1 and MAESTRO-NASH-1 which will read out in October.
Late-Breaking Oral Presentation: “Primary data analyses of MAESTRO-NAFLD-1, a 52 week double-blind placebo-controlled phase 3 clinical trial of resmetirom in patients with NAFLD” (LB005)
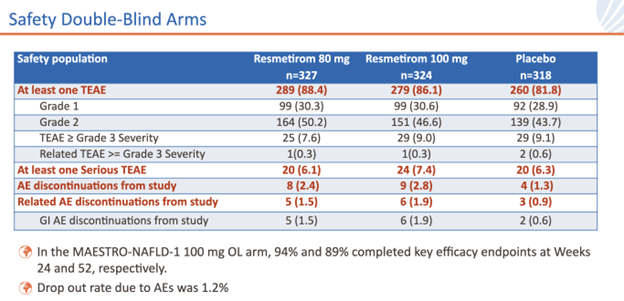
Primary and key secondary endpoints from the double-blind, placebo-controlled, 969-patient MAESTRO-NAFLD-1 safety study were achieved.
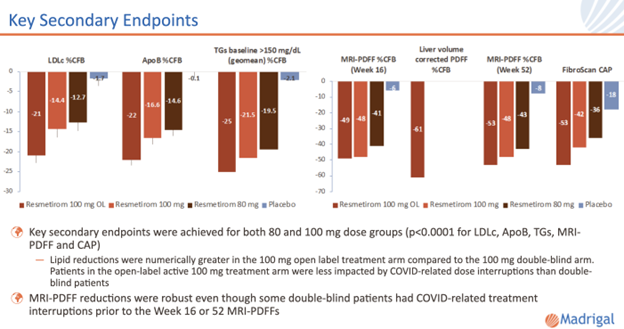
Resmetirom was safe and well tolerated and provided significant reductions in liver fat (measured using magnetic resonance imaging proton density fraction (MRI-PDFF) and FibroScan controlled attenuation parameter (CAP)), LDL-C, and other atherogenic lipids vs placebo.
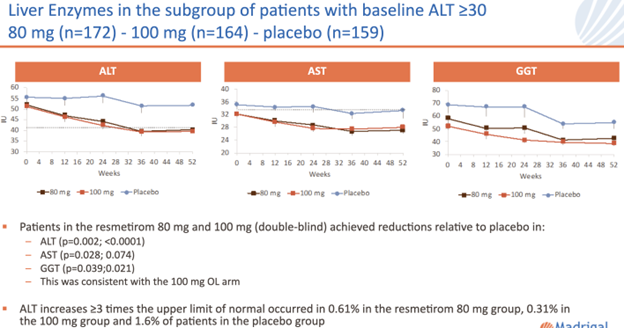
Patients treated with resmetirom also achieved significant reductions relative to placebo in liver enzymes ALT, AST, and GGT. This is another important safety advantage for resmetirom as reduced liver enzymes are a potential sign of liver disease improving and certainly better than elevated liver enzymes.
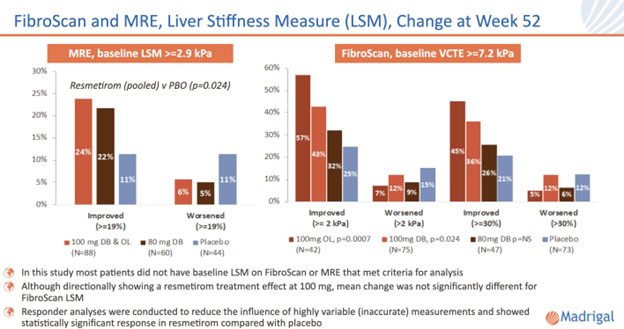
For those patients with sufficient baseline liver stiffness, as measured by FibroScan vibration-controlled transient elastography (VCTE) or magnetic resonance elastography (MRE), responder analyses showed statistically significant VCTE and MRE responses in the resmetirom groups compared to placebo.
Adverse event-related withdrawals were uncommon in the MAESTRO-NAFLD-1 study. The most common adverse event reported with greater frequency in the resmetirom groups vs placebo was generally mild diarrhea or increased stool frequency at the beginning of therapy.
Oral Presentation: “Biomarkers, imaging and safety in a well-compensated NASH cirrhotic cohort treated with resmetirom, a thyroid hormone receptor beta agonist, for 52 weeks” (OS121)
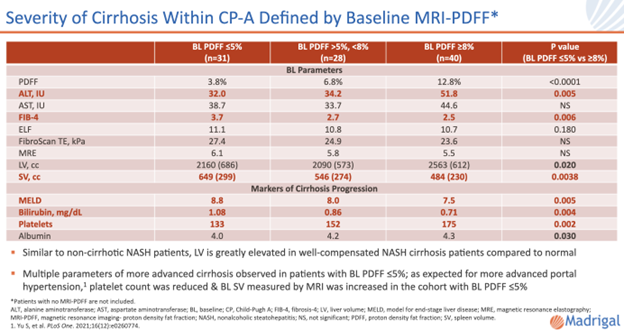
105 patients with well-compensated NASH cirrhosis were enrolled in the open-label arm of the MAESTRO-NAFLD-1 study. Baseline FibroScan VCTE (kPa 24.6) and MRE (5.7) scores were consistent with F4 fibrosis. Patients with lower MRI-PDFF (≤5%) at baseline had more progressed cirrhosis and greater spleen volumes. Similar to patients with non-cirrhotic NASH, liver volume was greatly elevated compared to normal at baseline.
Resmetirom reduced MRI-PDFF and LDL-C and other atherogenic lipids in patients with NASH cirrhosis and reduced FibroScan controlled attenuation parameter (CAP), VCTE, and MRE in a significant fraction of patients. The largest reduction in FibroScan VCTE (mean reduction of 9 kPa) occurred in the more advanced group (baseline PDFF ≤5%). Similar improvements were observed in MRE.
73% of patients, independent of baseline cirrhosis severity, had at least 15% reduction in liver volume at Week 52. Spleen volume was also reduced and was strongly correlated with liver volume change and exposure to resmetirom. Reductions in liver enzymes and atherogenic lipids were similar across patient subgroups.
Resmetirom was safe and well tolerated. As observed in patients with noncirrhotic NASH, mild GI adverse events were seen at the beginning of therapy. No differences in safety parameters between patients with cirrhosis compared to noncirrhotic NASH patients were noted. No thyroid axis changes or hyper- or hypothyroid symptoms were observed.
MDGL held a two hour conference call for investors after all of their presentations at EASL to discuss the outstanding resmetirom data presented at EASL yesterday. The Company focused on the 969-patient MAESTRO-NAFLD-1 safety study which showed the drug development candidate to have an excellent safety profile along with excellent efficacy and the 105-patient open-label trial in well-compensated cirrhosis patients which showed an excellent safety profile and both reduced liver and spleen volume. The two trials above combined with the soon to read out MAESTRO-NASH-1 Phase III trial mean that MDGL will have tested resmetirom in chronic liver disease patients at three different stages of disease; NAFLD patients are the least sick, followed by NASH patients, and compensated cirrhosis patients being the sickest. This is far and away the most comprehensive drug development program in the chronic liver disease space and bodes very well for the first FDA approval and then broad market penetration by resmetirom.
Importantly, MDGL continues to use every non-invasive means possible to measure for liver disease including routine blood tests and various types of MRIs. This is the “real world” treatment of chronic liver disease which rarely requires a liver biopsy. Current FDA guidelines require liver biopsies for NASH approval and MDGL has complied despite their modest use in “real world” diagnosis and treatment of NASH/chronic liver disease. MDGL will file resmetirom for FDA approval in early 2023 with close to 2,000 treated patients from two Phase III trials; MAESTRO-NAFLD-1 and MAESTRO-NASH-1 which will read out in October.