
June 16, 2025
BIOINVEST NEWS: Celldex (CLDX)
Celldex Therapeutics (CLDX – $21) – EAACI Update Further Strengthens Barzo’s Best-In-Class Profile — BUY
Celldex delivered another unprecedented clinical update for barzolvolimab (“barzo”) in CSU, further cementing the antibody’s position as the leading compound to change clinical practice in this disease of suffering. After the close on Thursday the Company reviewed the 76-week data of barzo demonstrating incredible durability in patients off-drug for 7 months showed complete disease control in almost half otherwise subjects. The data was presented at the EAACI conference in Glasgow on Friday. Then on Saturday, remarkable data in same trial in the most painful symptoms of CSU – angioedema – showed that more than three quarters of parents in the study were angioedema free after one year. In our view, both the likelihood of Phase III success and blockbuster potential of barzo is become more evident than ever. BUY
Off Drug Barzo Data Better Than Almost Every Other Drug Around
With remarkable one year (i.e., 52 week) data in CSU, at the EAACI conference this weekend CLDX presented results from patients in the study after being off the drug for 7 months. In what is the most severe patient population treated in any CSU study – the barzo study enrolled treated patients with up to 4x label dose of antihistamine (vs. xolair/ remi pivotal trials only up to 1x), ~65-75% were UAS7, ~65-80% had angioedema and ~20% had prior xolair exposure at baseline. Roughly 70% pts had severe CSU (UAS7≥28). With exceptional compliance for a >1 1/2 year study in chronic disease, 94% patients completed 76W from 52W (n=161/ 151 completed W52/ W76; 3 due to loss of efficacy, rest due to logistics etc.).
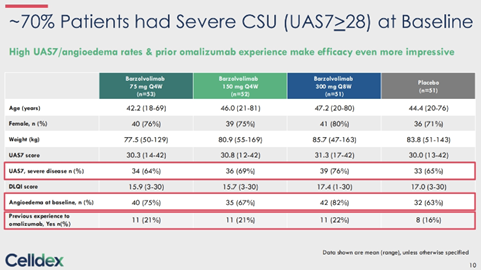
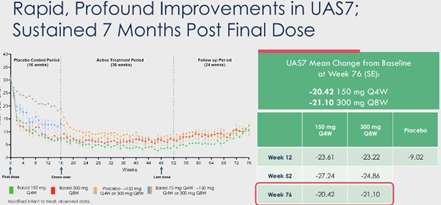
At the 7 months mark post-treatment, barzo’s unprecedented durability remains exceptional – complete response (UAS7=0) was 41%/ 35% (150mg Q4W/ 300mg Q8W) at W76, vs. 71%/ 52% at W52, and 51%/ 38% at W12 (vs. 6% by placebo). UAS7 change from baseline was -20.42/ -21.10 at W76, vs. -27.24/ -24.86 at W52, and -23.61/ -23.22 at W12 (vs. -9.02 by placebo). Well-controlled disease (UAS7≤6) was 56%/ 47% at W76, vs. 74%/ 68% at W52, and 60%/ 63% at W12 (vs. 13% by pbo). Serum tryptase at W76 was comparable to HV, and remained lower vs. baseline at both regimens, despite the fact that barzo was fully cleared at 24W after last dose. Patients quality of life (QoL) – of which Dr. Metz the lead investigator says may be the most important endpoint in CSU – QoL (DLQI score) 48%/ 40% (150mg Q4W/ 300mg Q8W) of treated subjects said that CSU had no longer any impact on their QoL at W76, vs. 82%/ 72% at W52, and 67%/ 57% at W12 (vs. 10% by placebo).
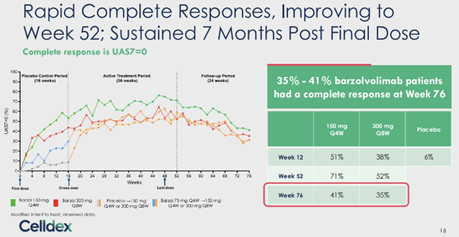
Note, CR is highly correlated with a meaningful QoL improvement, and recommended by current clinical guideline as the tx goal.
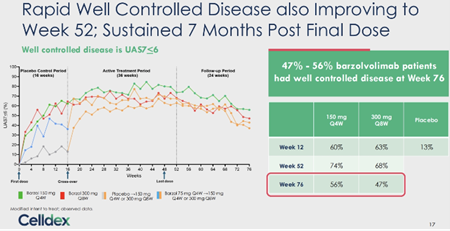
Dosing – With the Phase III loading doses, the Company and we believe that similar activity between 150mg Q4W and 300mg Q8W. (And efficacy may be even higher than the Phase II as well.)
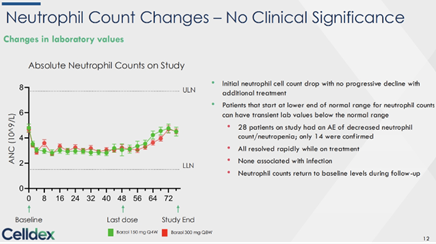
Safety – Small percentages (~10% +/-) of minor side effects (hair color changes, mild neutropenia and hypopigmentatio) have kept CLDX stock under pressure. In this post-treatment profile safety signals were reversed in the vast majority of cases, with whatever remainder also correcting and likely to be cleared over the next month or so (according to investigators). Dr. Metz pointed out that no one complained or left the study due to these mild A/Es – because their disease was gone. (The only one to complain was non-sophisticated investors – sorry but true) The drug’s safety show with no new signals, and neutrophil count changes/ hair color change/ skin hypopigmentation all reversed over the off-Tx follow up. Neutrophil count changes initial drop was observed no progressive decline after first few weeks (and steadily improved) and corrected during the off-drug period (as management always believed happens but got no credit – until now). As a reminder infections were similar on both drug and placebo arms – suggested no issues.
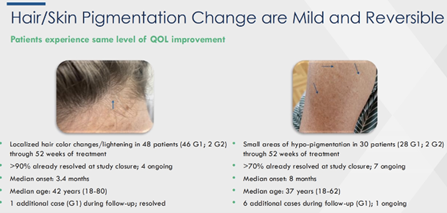
Hair color change localized change/ lightening were reported in n=48 patients (46/2 G1/G2) through W52, 90%+ of which resolved at study closure and n=4 ongoing. 1 addtl case (G1) during followup and resolved. Median time to onset/ resolution was 3.4/ 5.6 mo. Skin hypopigmentation was reported in n=30 pts (small area, 28/2 G1/G2) through W52, 70%+ of which resolved at study closure w/ n=7 ongoing. 6 addtl cases (G1) during follow up with just 1 ongoing. Median time to onset/ resolution was 8/ 7.3 mo. Co notes anecdotal improvements from physician.
CONCLUSION – New Standard of Care Coming – Along With Blockbuster Status in CSU Alone
By most estimates, CSU alone affects between 0.5-1% of the population. Hence, we believe the data show the very wide and widening therapeutic window of barzvolvolimab. Stock is vastly undervalued by this metric alone. In addition to the current Phase III trial in CSU, Celldex will shortly initiate a second Phase III trial in Chronic Inducible Urticaria (CindU). In addition, the company will present follow-up results from the Phase II CindU trial in H2:25.
Many other mast cell indications on the way – the most near-term is EoE. Likely in late-Q3 by our estimate, Celldex will release Phase II data in Eosinophil Esophagitis – another mast cell mediated condition (likely wrongly termed). Should that study prove successful, we believe the stock will begin to finally discount the myriad (up to 75) conditions that mast cell play a key role in – despite the fact that the Phase III trials will not read out until H2:26. Other Phase II studies under way include atopic dermatitis and prurigo nodularis.
